Sick Bees – Part 18F4: Colony Collapse Revisited – Environmental Toxins
Sick Bees Part 18f4: Colony Collapse Revisited
Environmental Toxins
First published in ABJ April 2013
Randy Oliver
ScientificBeekeeping.com
New Chemicals in the Environment
Fear of Chemicals – It Ain’t Just Pesticides
Irrational Fear of “Chemicals”
General Environmental Pollutants
Randy Oliver
ScientificBeekeeping.com
I left off last month with the analogy of the bee colony being akin to a leaky boat needing to “bail out” (detoxify) numerous plant alleleochemicals. Generally, they do a pretty good job at this. But what if we now add more “leaks” in the form of manmade chemicals?
New Chemicals in the Environment
The modern era of chemical pest control began around the time of World War II, when the synthetic organic chemical industry began to develop. The first synthetic organic pesticides were organochlorine compounds, such as DDT. At that time, DDT was considered to be a wonder of modern chemistry. It was cheap, knocked the snot out of insect pests, and appeared (at that time) to be relatively safe to humans. DDT and other organochlorine insecticides were widely used until scientists noticed that they were persisting in the environment (they did not readily degrade) and, more seriously, could bioaccumulate in birds, humans, and other animals. In 1962 Rachel Carson, in her book Silent Spring, introduced the term “ecosystem” to the general public, and kick started the environmental movement. As a result, the EPA was created in 1970 [1], which subsequently banned most of the organochlorine pesticides [2].
Up until Silent Spring, “chemicals” had a good name, synonymous with “progress” (Fig. 1). The heyday of modern chemistry ran from WWII through the 1980’s, during which time the number of newly-created chemicals grew exponentially [3].
Figure 1. Americans have gone from embracing “chemicals” to now being fearful of them. Although I certainly wish to see chemical pollution of our air, water, and landscapes cleaned up, and am cautiously concerned about any manmade chemicals in my diet, I feel that public sentiment may have swung too far, based upon ignorance and unwarranted fear.
Chemists today have gone full circle, and now are at the forefront of analyzing and explaining how any pesticide or pollutant can affect health or ecosystems [4]. Unfortunately, the public confuses our newfound ability to detect chemicals in the parts per trillion with increased risk– a major misunderstanding which frustrates both toxicologists and the EPA risk assessors [5].
Scientific note: one thing that bugs me about studies on the sublethal effects of synthetic pesticides is that we have no perspective as to the degree of impact due the pesticide relative to the adverse effects of common plant alleleochemicals. I’d like to see some benchmarking of a few common plant phytotoxins that could then be run as “positive controls.”
Fear of Chemicals—it ain’t just pesticides
We live in a society obsessed with fear. These days you can’t even find a merry-go-round or jungle gym on a playground any more for fear that some child might get hurt while playing outdoors! And in my chemical-phobic home state of California, every McDonalds and Starbucks are required by law to post warnings that French fries and coffee contain acrylamide–a lethal neurotoxin, carcinogen, and reproductive toxicant [6]. At the lumber yard, my receipt now warns me that:
WARNING: Drilling, sawing, sanding or machining wood products generate wood dust, a substance known to the State of California to cause cancer.
For Pete’s sake, coffee, French fries, and sawdust are now considered to be “substances” that can cause cancer! Heaven forbid that they ever get hold of my smoker!
Irrational Fear of “Chemicals”
The term “chemical” has become emotionally loaded. In truth, there is nothing inherently good or bad about “chemicals”–everything that you can touch, taste, or smell is a chemical. And as Paracelsus pointed out 500 years ago, “All things are poison, and nothing is without poison; only the dose permits something not to be poisonous.”
How about that proverbial grain (or teaspoon) of salt with which we should take the alarmists’ warnings? Salt is an effective and nonspecific poison–1 tsp of salt is about 1/10 the lethal human dose [7]; salt is also mutagenic, can cause reproductive problems and muscular dysfunction. Even water or oxygen can kill you if you consume too much. It is all about the dose!
And don’t let multi-syllable names scare you. Consider beta-dextro-fructofuranosyl-alpha-dextro-glucopyranoside. This chemical is arguably the leading cause of preventable death in the U.S.–strongly linked to heart disease (the #1 cause of death), Type 2 diabetes, obesity, hypertension, colon cancer, and tooth decay [8], yet there are people who feed it to their children! I’m sure that you’ve figured out its common name by now—table sugar.
Manmade Chemicals
I’m going to get back to pesticides soon, but first we should realize that our ecosystem is full of plenty of manmade chemicals other than pesticides—a number of which should be considered when we are looking at the background level of toxins that the bees must deal with (as per my leaky boat analogy).
General Environmental Pollutants
Long-time bee researcher and toxicologist Dr. Jerry Bromenshenk points out that our focus on agricultural pesticides overlooks plenty of other toxic substances to which bees are exposed (which got plenty of attention from beekeepers prior to the distraction of synthetic pesticides). His research suggests that we could consider bees as “flying dust mops,” and hives to be “air sampling devices.”
Bromenshenk, funded at the time by the EPA, developed methods for using beehives as monitors of environmental pollution. In 1989, based on his studies, EPA approved honey bees in their guidance reference manual for establishing and conducting ecological assessments of hazardous waste sites [9]. I’ve condensed his findings (from the book Honey Bee: Estimating the Environmental Impact of Chemicals [10] and personal communications):
A colony of honey bees is an effective environmental sampling device for volatile and semi-volatile organic compounds. Beehives located in uncontaminated environments contain compounds released by the bees themselves, from hive stores, and from the materials from which the beehives are constructed. In all areas they also contain compounds from vehicles, farms, industries, and households in the hive vicinity. Some of these are pesticides, but we also identified on average more than 200 other (volatile and semi-volatile) chemical compounds that occur simultaneously inside each and every beehive, as well as trace elements, heavy metals, and even radioactive materials, sometimes at levels that caused bee toxicity or queen mortality. Thus, these chemicals should be included in any discussion of honey bee health.
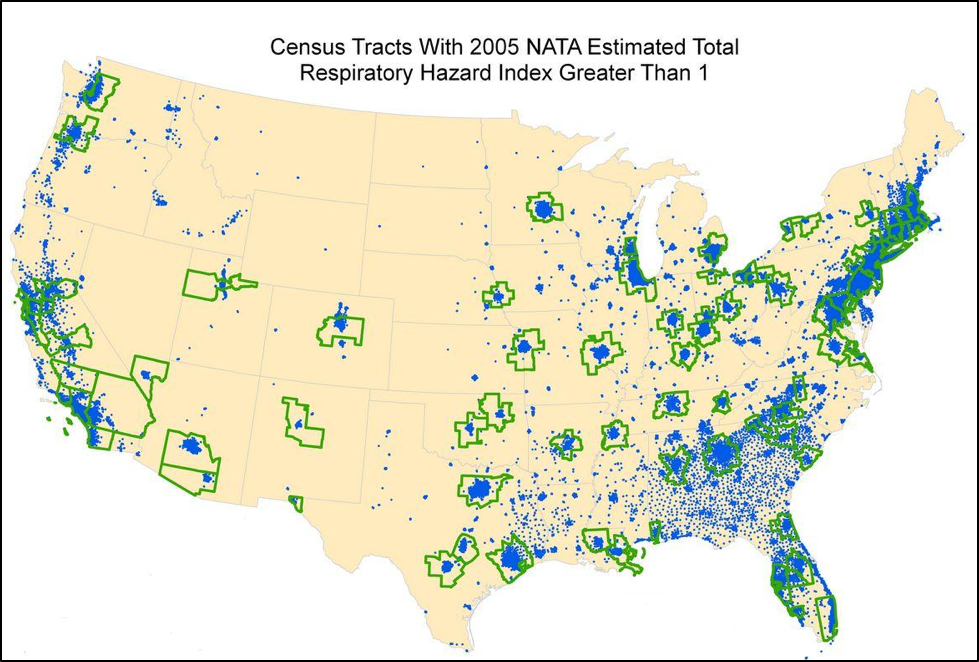
To our credit, we have cleaned up pollution significantly since the ‘70’s, but there are still plenty of dusts, solvents, and volatile emissions that can be carried back to the hive by foragers (Fig. 2).
Figure 2. The EPA tracks the levels of 177 air pollutants such as acetaldehyde, arsenic, mercury, benzene, carbon tetrachloride, formaldehyde, methyl chloride, toluene, and several pesticides [11]. Dusts stick to bees due to electrostatic charge, and are carried back to the hive, where they are inadvertently mixed with the pollen in the beebread. Volatiles accumulate and may even bioconcentrate inside the bodies of bees and may be absorbed by the beeswax as bees ventilate the hive.
Bromenshenk pioneered using bee hives to track the distribution of trace element pollution downwind from smokestacks. Oddly, his seminal paper [12] is widely cited in environmental studies, but rarely in the bee literature! He also demonstrated that such heavy metal pollution could show up in hives even decades after a smokestack was shut down, and that such pollution could cause colonies to decline in strength [13]
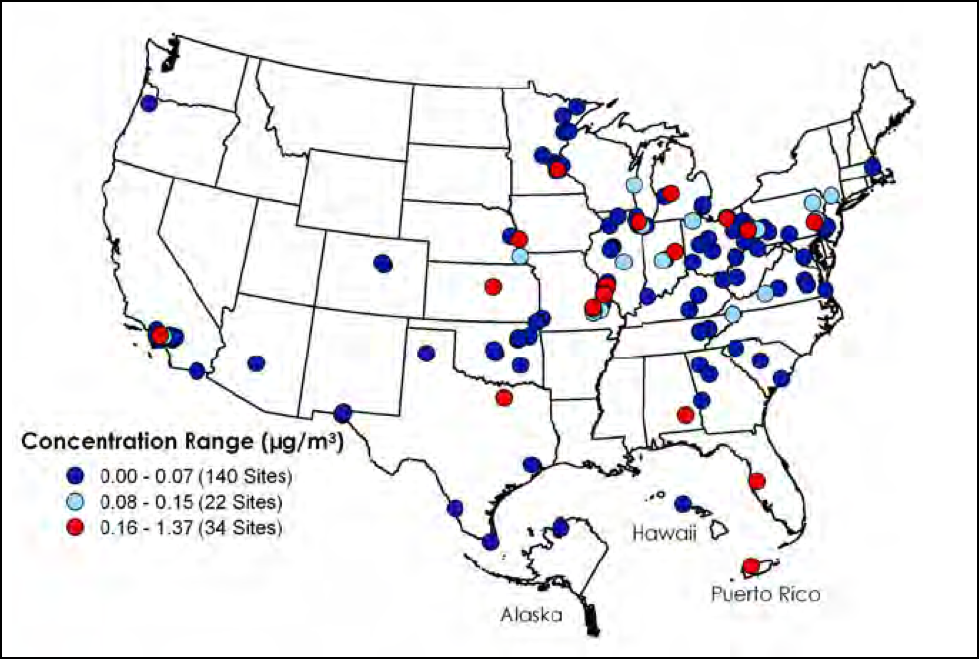
Again, although we have made progress in reducing smokestack emissions, there is still plenty of heavy metal pollution going on (Fig. 3). Keep in mind that unlike pesticides, heavy metals never degrade, remaining in the soil forever, possibly being concentrated in the pollen of plants growing there. For example, lead-arsenate insecticides were widely used on cotton until they were banned in the ‘80’s. But they’ve come back to haunt us when rice is then planted on previous cotton land [14].
Figure 3. Heavy metal pollution is invisible. The above map shows the amount of lead in the air in 2010 (in μg/m3; maximum 3-month averages). Some plants exhibit the nasty habit of bioaccumulating heavy metals in their pollen [15]. Source EPA [16].
***
Update March 2015 One natural trace mineral in pollen is manganese (which may be elevated in areas of high manganese content of the soil). Manganese is also an air pollutant released by vehicle emissions, industry, and iron smelters. A team of researchers found “that manganese exposure negatively affects foraging behaviour in the honeybee…we found that honeybees treated with 50 mM Mn2+ showed a precocious transition from in-hive behaviours to foraging … Surprisingly, precocious foragers completed significantly fewer foraging trips over their lifetime …, which suggests that long-term exposure of beehives to Mn2+ could negatively affect colony fitness.
Søvik, E, et al (2015) Negative impact of manganese on honeybee foraging. Biology Letters http://rsbl.royalsocietypublishing.org/content/11/3/20140989
For releases of manganese in your state, see http://www.atsdr.cdc.gov/toxprofiles/tp151-c6.pdf
***
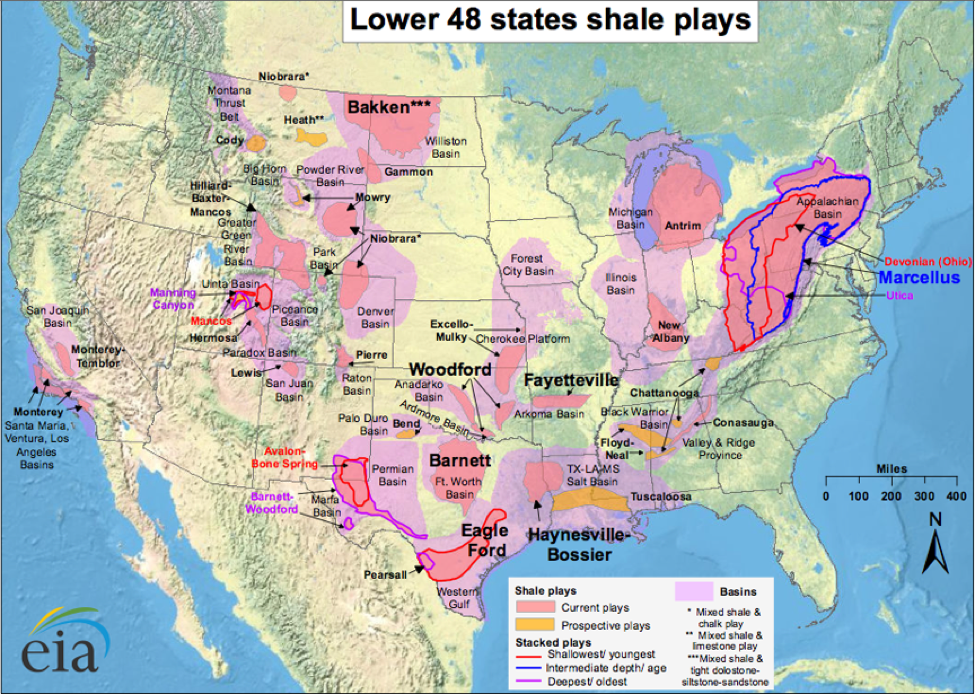
And now I wonder about the volatiles being released by the fracking of shale formations—common in many beekeeping areas (Fig. 4).
Figure 4. Fracking releases a number of potentially toxic petroleum hydrocarbons into the air, including benzene, ethylbenzene, toluene and xylene [17]. Do you place any hives near oil or gas wells? Source [18].
Bromenshenk points out that when looking for the causes of bee health issues, we should keep in mind the background levels of environmental pollutants, which to my knowledge, are rarely tested for.
Practical application: Do you know the history of heavy metal pollution where you place your bees? If there was ever mining, a factory, a smokestack, or use of lead-arsenate insecticides, the soils and plants in that area may be quietly poisoning your bees and you’d never know!
Scientific application: Densely populated urban areas tend to have high exposures to a vast array of pollutants as evidenced by the preceding maps. This fact should be taken into account in any trial of pesticide effects performed in such areas.
Natural Elements
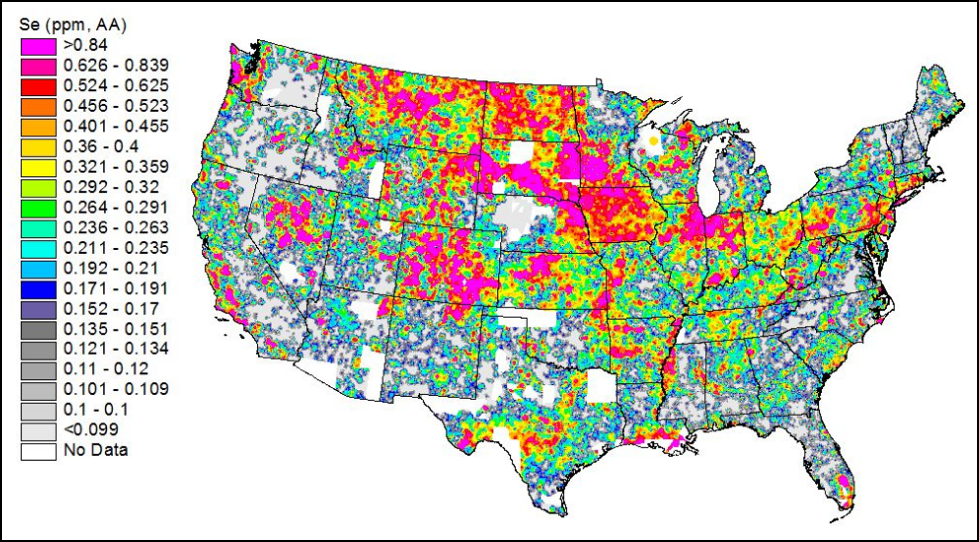
So far, I’ve been speaking of manmade pollutants. But some plants concentrate natural soil elements to high concentrations in nectar and pollen [19]. Beekeepers should be aware of a couple of recent studies on selenium and bees. Quinn [20] found that some plants concentrate selenium in nectar and pollen to levels that could be toxic to bees, and that surprisingly, bees do not avoid those flowers! Hladun [21] then demonstrated that such naturally-occurring levels of selenium can cause the same sorts of behavioral and mortality effects upon bees as one would see in the standard testing of insecticides!
Practical application (?): commercial beekeepers often summer bees in areas of high soil selenium (Fig. 5). Some native plants in those areas concentrate selenium to toxic levels. Of special concern is that alfalfa and canola can also concentrate selenium.
Figure 5. Map of selenium concentration in soils. Bees kept in the reddish areas stand the risk of being exposed to toxic levels of this metal via nectar and pollen, especially in drought years. Source [22]. You may wish to view a similar map for the arsenic content of soils [23]!
And if unavoidable environmental pollutants weren’t enough, bees have the annoying habit of getting “into things.”
Those Danged Bees!
Not only do bees get into soda cans and any other source of potentially harmful sweets (like cracked fruit, molasses, or ant or fly bait), but they are programmed to seek out toxic plant resins to use as propolis (Fig. 6).
Figure 6. I’ve heard from two different friends that when they pull into certain trailer parks, that bees come and scrape the caulking out of the seams of their travel trailers (note the caulking in the pollen baskets). In both cases the bees found the caulking of only one particular brand of trailer in the entire park to be attractive! I have no idea as to the toxicity of the caulking once back in the hive. Photo courtesy Kerry McDonald.
Poor Colony Survival
It’s easy to blame poor colony performance at the end of the season on pesticides, but are they always truly to blame? I took the photo below (Fig. 7) in late July—about a month after the main honey flow ended.
Figure 7. This colony appears to be well fed—notice the distended abdomens full of nectar and the abundant beebread. However, if you look closely at the young larvae, you’ll notice that they are only being given a minimum amount of jelly, and the brood pattern is shot (indicating poor larval survival). Despite the presence of pollen and honey, the signs are that this colony is under serious stress!
If you inspected the above colony in late summer, you’d see pollen stores, well-fed adults, and active broodrearing—yet it is clearly under severe stress. If the colony had previously been exposed to ag chemicals, the beekeeper might suspect that they were to blame.
However, the colony was one of mine, and I feel that I can safely assume that it had not been exposed to any pesticides (no crops or commercial landscapes within flight range). I have no idea as to whether the stressed brood was due to an inadequate nutritional profile of the late-season pollen, plant alleleochemicals, or something else. In my experience, this stressed colony (which is typical for my area at this time of year), without nutritional intervention by the beekeeper, would dwindle in strength, be unable to produce a healthy winter cluster, and likely not make it through winter, eventually succumbing to viruses or nosema. The question then would be to what the beekeeper attributed that loss. In this case, something sure wasn’t right, but it certainly wasn’t pesticides or miticide residues!
Practical application: colonies require high-quality pollen to prepare for winter. Those suffering from poor nutrition may not be able to rear a healthy crop of “winter bees” [24, 25]. Such nutritional stress can not only cause the population to dwindle, but such dwindling could be exacerbated by the effects of any toxins, natural or manmade. And an exposure to a dose of agricultural pesticides that a healthy colony might simply shrug off could be the kiss of death for such a nutritionally-stressed one!
Reality Check
Let me be clear that I’m not suggesting that pollution and plant alleleochemicals are the cause of most colony mortality, nor that pesticides aren’t involved. Rather, the point of the discussion is that the local background levels of environmental pollutants and plant allelochemicals may help to explain why study after study has been unable to find a statistical relationship between colony mortality and pesticide residues [26]—it’s hard to figure things out when you’re working half blind! The problem may be that there were additional variables that were not measured (each analyis adds to costs). I have yet to see a study in which colony mortality was investigated in the full context of the sum total of all toxins (including those from plants, pollutants, air, and dust) to which the colony was exposed—these additional toxins are often present at levels that rival the toxicity of pesticides! Such additional variables could easily confound attempts to link [27] colony mortality to pesticide residues.
Practical application: It is relatively straightforward to nail the cause of an acute pesticide kill. It is much more difficult to separate any sublethal effects due to pesticides from those of the stew of natural and manmade toxins to which bees are exposed in the real world. Unless we take into account all the leaks in the boat, we really don’t know how hard the colony is bailing to stay afloat. Colonies may be able to shrug off an exposure to an agricultural pesticide in one location or under certain conditions, yet suffer serious mortality in another location or under other conditions. Such variability makes it devilishly difficult to tease out the actual relationships between pesticide residues and colony health.
NEXT—on to (finally) manmade pesticides!
Acknowledgments
As always, I’m indebted to Peter Borst for his assistance in research, and to Drs. Jerry Bromenshenk and James Frazier for their helpful reviews and comments.
References and Notes
[2] Borek, V (2012) History of pesticides. (Broken Link!) http://www.chemistryexplained.com/Ny-Pi/Pesticides.html#ixzz2EzTpmYPS
[3] Pimentel, GC (1985) Opportunities in chemistry. http://www.nap.edu/openbook.php?record_id=606&page=4
[4] Laszlo, P (2006) On the self-image of chemists, 1950-2000. HYLE–International Journal for Philosophy of Chemistry 12(1): 99-130. http://www.hyle.org/journal/issues/12-1/laszlo.htm
[5] Gold, LS, et al (2001) Pesticide residues in food and cancer risk: a critical analysis. In: Handbook of Pesticide Toxicology, Second Edition (R. Krieger, ed.), San Diego, CA: Academic Press, pp. 799-843 (2001). http://potency.berkeley.edu/text/handbook.pesticide.toxicology.pdf I highly recommend this read!
[8] http://www.cdc.gov/PDF/Frequently_Asked_Questions_About_Calculating_Obesity-Related_risk.pdf
[9] Warren-Hicks, W, et al (1989) Ecological assessment of hazardous waste sites: A field and laboratory reference. EPA/600/3-89/013. A free download.
[10] Smith GC, GH Alnasser, DC Jones , and JJ Bromenshenk (2002) Volatile and semi-volatile organic compounds in beehive atmospheres. In, Devillers and Pham-Delègue, eds., Honey Bees: Estimating the Environmental Impact of Chemicals, CRC Press.
[11] http://www.epa.gov/ttn/atw/nata2005/05pdf/2005polls.pdf
[12] Bromenshenk JJ, et al (1985) Pollution monitoring of Puget Sound with honey bees. Science 227(4687): 632-634.
[13] Bromenshenk, JJ, et at (1991) Population dynamics of honey bee nucleus colonies exposed to industrial pollutants. Apidologie 22 (4) 359-369.
[15] Roman, A (2009) Concentration of chosen trace elements of toxic properties in bee pollen loads. Polish Journal of Environmental Studies 18(2): 265.
[18] Energy Information Administration.
[19] Terry, N, et al (200) Selenium in higher plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 51:401–32. http://www.plantstress.com/articles/toxicity_i/selenium.pdf
[20] Quinn CF, et al. (2011) Selenium accumulation in flowers and its effects on pollination. New Phytol 192: 727–737. http://rydberg.biology.colostate.edu/epsmitslab/Quinn%20et%20al%202011%20New%20Phytol%20proofs.pdf
[21] Hladun KR, et al (2012) Selenium Toxicity to Honey Bee (Apis mellifera L.) Pollinators: Effects on Behaviors and Survival. PLoS ONE 7(4): e34137. http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0034137
[24] N. Höcherl, N, et al (2010) Effects of a pure maize pollen diet on the honeybee. Apidologie 41: 676–694. Free download.
[27] The other unknown variables (pollutants, alleleochemicals, nutrition, temperature) can bedevil the statistician when he tries to separate out the “effect” due solely to the pesticides. The researcher is trying to determine whether the “independent variable” (pesticide level) correlates with the “dependent variable” (whatever aspect of bee or colony health being measured. If there is a great deal of additional variation due to unmeasured factors such as pollution or plant allelochemicals, any sublethal effects due to pesticides may get buried in the “noise.”