2022 Extended-Release Oxalic Update: Part 1
April 22, 2022
2022 Extended-Release Oxalic Update
Part 1
Randy Oliver
ScientificBeekeeping.com
First published in ABJ March 2022
As more and more beekeepers try to wean themselves off amitraz, there is great interest in oxalic acid. Unfortunately, a dose of OA by any of the currently-approved application methods exhibits little residual action, so has limited efficacy if a colony is rearing brood. However, as with other miticides sold in extended-release strips or formulations, oxalic can be applied in a similar manner.
REVIEW OF MY FINDINGS ON EXTENDED-RELEASE OXALIC ACID (OAE) TO DATE
My sons and I have experimented, by permit [[1]], with OAE for some time, testing a range of ratios of oxalic to glycerin, as well as with a variety of delivery matrices. So far, we’ve determined that:
- We like the handling characteristics and efficacy of the 1:1 (by weight) ratio.
- Swedish sponges make a great delivery matrix, but are pricey.
- OAE has the potential to take mite infestation rates down to zero, but it is a long-term treatment. It doesn’t cause as rapid a reduction of mite levels as do most other treatments (formic is the quickest). It requires about two months to exhibit full efficacy.
- Very importantly, due to its slow action, highest efficacy is attained in yards where there is little mite immigration.
- Its efficacy does not appear to be affected by temperature or humidity (since broodnest environment is regulated by the bees).
- It’s best applied proactively (early in the season), rather than reactively (waiting until the mite level gets high).
- It’s easy on the bees and brood.
- It’s not a worry as far as contamination of honey or the combs.
- Unfortunately, this application method is not yet approved by EPA, and I in no way encourage or endorse the use of it without a proper permit.
OAE HOLDS GREAT PROMISE
Although this application method of oxalic acid is not yet approved by our slow-moving EPA [[2]], it is clear to me that it is going to be a game changer for varroa management. My sons and I are impressed by how it has performed in our own authorized experiments. I also get reports from beekeepers in countries where it is approved or allowed [[3]], as well as from beekeepers elsewhere (Figure 1).

Fig. 1 Beekeepers from all over the world send me glowing reports on how well OAE can work. The photos above are from a professional East Coast beekeeper (whom I assume is permitted), who waxed poetic about how OAE treatment resulted in the strongest, healthiest, and most honey-productive colonies in memory, holding the mite levels in his busting colonies to low levels. His photos speak for themselves.
Practical application: I’m not trying to sell or promote this application method (especially since it’s not yet approved). It’s not a panacea, and is best used proactively. And as with other treatments, there are always outlier colonies in which mite levels somehow don’t respond, and the treatment may not be able to keep up with heavy mite immigration.
But because it would fit very well into our own commercial operation (since we eschew comb-contaminating synthetic miticides), my sons and I continue to perform permitted research not only for the benefit of the beekeeping industry, but also in own interest — and for that of our poor bees, who beg for relief from this parasite. In the long run, OAE may help us to stave off varroa until mite-resistant bees become widely available [[4]].
TESTING VARIOUS MATRICES
The concept of OAE is to provide sustained release — from an inert delivery matrix — of the oxalic acid onto the bees. Glycerin acts as the solvent and carrier of the acid, and also has the advantages of being inexpensive, food-grade, a humectant (which helps the acid to be effective), an adhesive (that helps the acid to stick to the bees), and has the added benefit of being distasteful to the bees (so they’re less likely to consume the acid). But what’s the best matrix to use?
I’ve scoured catalogs and stores for suitable absorbent sheets or pads (preferably biodegradable), and gotten suggestions from beekeepers everywhere. This spring I chose seven matrices to compare to the successful Swedish sponges (which work better than do the shop towels and cardboard that I’d previously experimented with).
I had purchased a number of different brands and shapes of cotton wipes and pads to try, and decided to use the Q-tips brand, since it had crimped edges. Because they happened to be round, for comparative testing I measured their surface area (4 in2), and cut rectangular strips of identical surface area from the other matrices.
I prepared six pads of each matrix, using a single batch of OA/gly solution, and then allowed them to come into equilibrium at a fixed temperature and humidity in an incubator. After weighing the pads, beekeepers Brion and Alice Dunbar helped me to haphazardly place one pad of each matrix into each of six hives (Fig. 2), and allowed the bees to walk over them for 20 days (7 April to 27 April).

Fig. 2 In order to determine how well each matrix dispersed the OA/gly solution onto the bees, we covered the top bars of the lower brood chambers of six hives with masking tape to prevent absorption, and weighed the saturated strips before and after 20 days in the hive. The yellow-starred matrices looked to be the most promising.
We then collected the pads, again allowed them to come into equilibrium in the incubator, and reweighed them as a group in order to determine the average amount of weight lost per pad (Table 1).
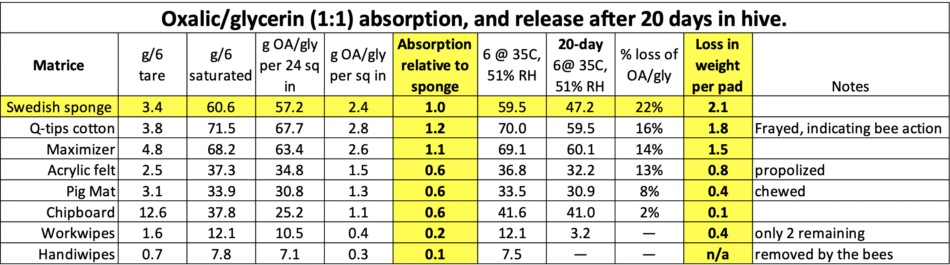
Table 1. The tested matrices, ordered by weight loss. None lost as much as the sponges, but some were close. I’ve highlighted the most important cells.
Three of the matrices showed promise, due to their amount of absorption or release, as well as other characteristics (the lowest two had been removed by the bees). So I made up a batch of them, along with some sponges (Figure 3).

Fig. 3 I prepared the four matrices to be tested with the same batch of OA/gly solution at the 1:1 ratio. Tip: for full absorption, it’s important to give any matrix enough room to expand.
I then ran a quickie trial of the four, testing them by seeing how much they increased the rate of mite drop after application (in 2 colonies for each matrix, randomly blocked by starting mite count) (Figure 4).
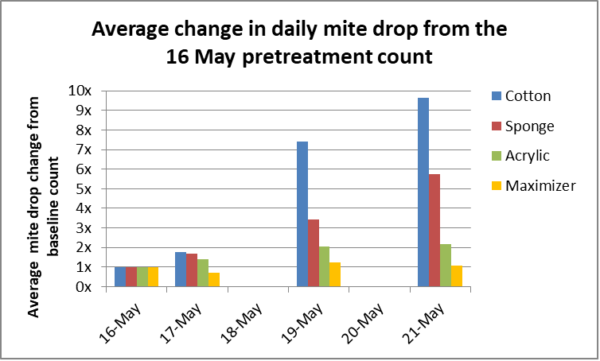
Fig. 4 In my preliminary trial of the four most promising matrices, I was unfortunately only able to track mite drops for five days (16-May was the pretreatment baseline count). The cotton pads caused by far the greatest short-term increase in mite drop.
Wow, cotton pads looked really promising! I had hoped that the bees might tug the saturated cotton fibers off, and drag them down through the colony (Figure 5).

Fig. 5 The above photo was taken after the pads being in the hive 10 days. There didn’t appear to be the amount of tugging of the fibers that I’d hoped for. Note the slight propolization around the edges.
Unfortunately, the facial pads were difficult to separate after preparation, and would require 13 round pads to equal the surface area of a single Swedish sponge. So I didn’t follow up any more with them this season.
Practical application: Cotton in some form still remains a very promising candidate (I plan to try terrycloth or other absorbent fabric, and plastic-free feminine-hygiene pads). I also experimented with adding thymol to the pads, but it didn’t appear to improve performance. Properties to consider with any matrix are its handling characteristics, including preparation, field application, and ease of removal from the top bars after treatment.
SOME QUESTIONS TO ANSWER
As I reported last year [[5]], we had, in late spring and early summer, applied a quarter-sponge strip of OAE to a large number of colonies growing from nucs in experimental hives in several yards, and found that a minimal-dose treatment largely suppressed increase in mite wash counts for a few months. So we were curious as to whether such a low-dose treatment would prove to be efficacious in larger low-mite colonies. And based upon our preliminary experimentation with the alternative matrices above, I thought that we could run a single comparative field trial to answer three questions:
- How do Swedish sponges, Maximizer pads, or acrylic felt compare in efficacy at reducing mite loads?
- Is there a minimal surface area (as opposed to amount of OA) of OAE pads necessary to obtain good efficacy in double deeps?
- Can you dose with OAE proportional to the mite infestation rate? e., can you use fewer strips in a low-mite colony?
For experimentation, we had set aside a bunch of intentionally-untreated colonies with 2nd-year queens, grown from March and April nucs split from colonies returned from almond pollination, in four different yards. So in late May — as their mite infestation rates climbed — I thought up an experimental design to attempt to answer the questions above.
A LARGE-SCALE FIELD TRIAL
Last year we learned that one half-sponge reduced mite levels substantially, and two half-sponges exhibited very good efficacy. I figured that if we cut the pads into strips the same size as a quarter of a Swedish sponge, and then apply only a single strip to the lowest-mite hives, going up to 4 strips for the highest-mite ones, that we could perhaps fine-tune the optimal dose.
We started the trial by taking mite wash counts from all the hives in the yards on May 20-21, sorted them by starting mite level, and randomly assigned treatment types accordingly (Table 2).
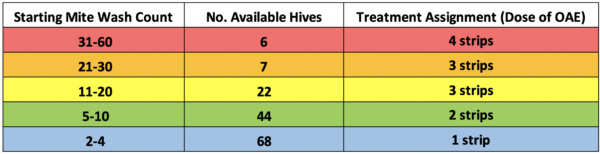
Table 2. We unfortunately didn’t have many colonies with mite counts above 30, and in retrospect, I should have assigned the 4-strip treatment to more colonies in the 11-30 mite groups.
Scientific note: We randomly assigned the treatments, blocked by starting mite count. We didn’t run any negative Control (untreated) groups, since we were testing for comparative, rather than absolute efficacy (and it would have taken eight Control groups). If you look at the dates of all these experiments, you can see that we were performing experiments back-to-back in short order, rushing against the season and mite buildup. I had to make quick, on-the-spot decisions, and in retrospect sometimes wish that I had done something differently.
We applied the strips on May 26-27 (Figure 6). On June 30 we spot checked colonies for strip condition and mite wash counts. At that time point, the acrylic felt did not appear to be performing, so we removed those colonies from the trial.

Fig. 6 According to the starting mite wash count, we applied from one to four 1¾ “ x 7½ ” strips per hive after smoking the bees off of the top bars.
On July 20 (after 55 days of treatment) we took ending mite wash counts.
RESULTS FOR THE SWEDISH SPONGES
The results are shown in Figures 7 and 8 — the more blue you see relative to red means the more efficacious the treatment.
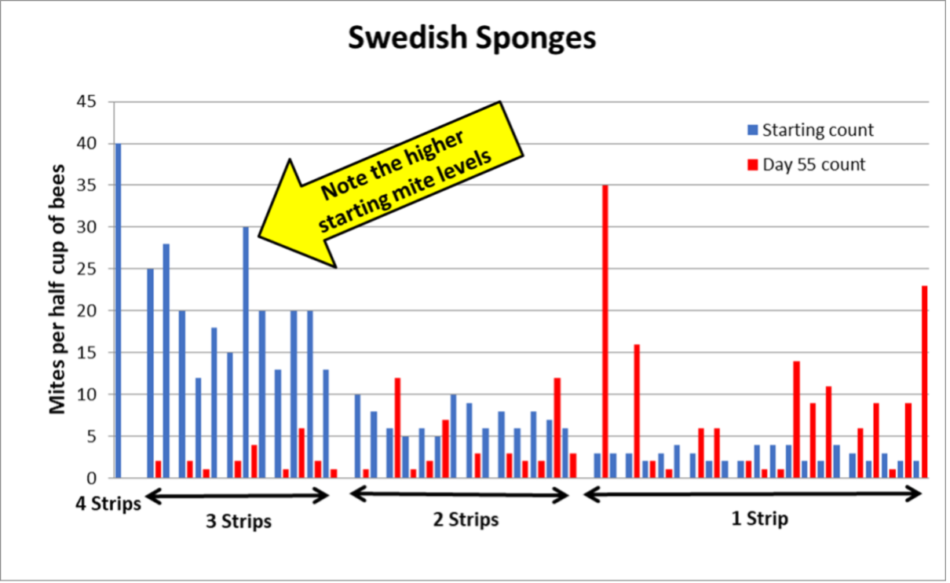
Fig. 7 Contrary to our observations the previous year, applying only a quarter sponge to low-mite colonies did not do the trick (too many had their infestation rates increase substantially). But similar to last year’s results, two strips (a half sponge equivalent) reduced the mites, and a full-sponge equivalent (4-strips) nailed it. Note that some of the highest-mite hives went to counts of zero.
Practical application: When I shared the results with my sons, it was clear that a 4-strip equivalent (two half-sponges) would now be our standard OAE treatment for full-strength colonies.
RESULTS FOR THE MAXIMIZER PADS
The results for the Maximizers were quite similar to those for the Sponges (Figure 8).
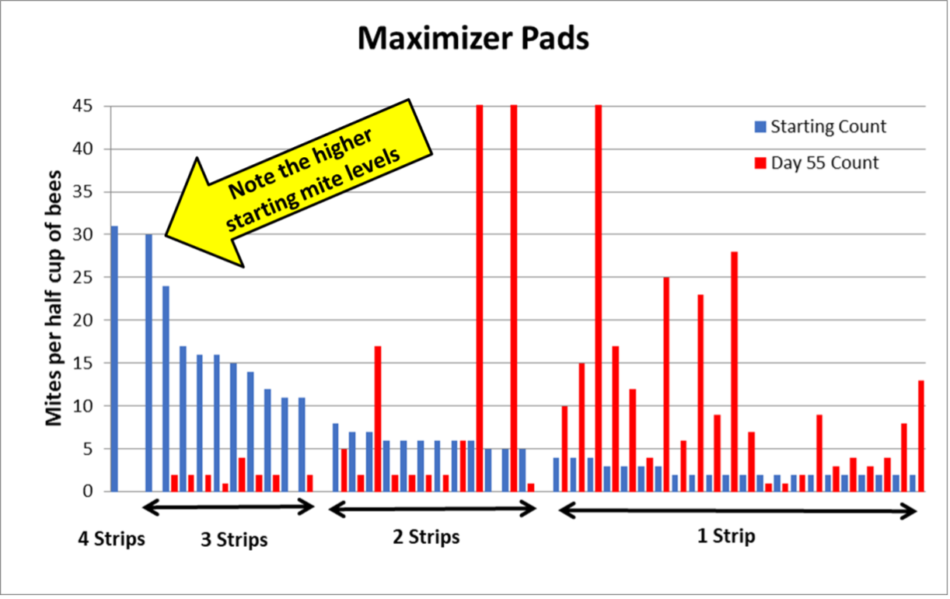
Fig. 8 It looks as though the Maximizers may be slightly less efficacious than the sponges at low dose. As with with the sponges, the 4-strip hive zeroed out.
Practical application: Once a colony’s virus infection gets too high due to having an elevated mite load, it may not be able to be saved by any treatment. I’ve been surprised that in our previous trials, virtually all high-mite colonies recovered if given an OAE treatment. In this trial, one of the two highest-mite 4-strip colonies for each matrix type did not make it, but the other one not only survived, but ended up with a mite count of zero!
MITE REDUCTION AND EFFICACY CALCULATIONS
Let’s take a look at the comparative effect of treating with either 1 or 3 strips of the two matrices. To do so, I simply totaled the mite counts for all the colonies in each treatment group at the start and end points (Figure 9).
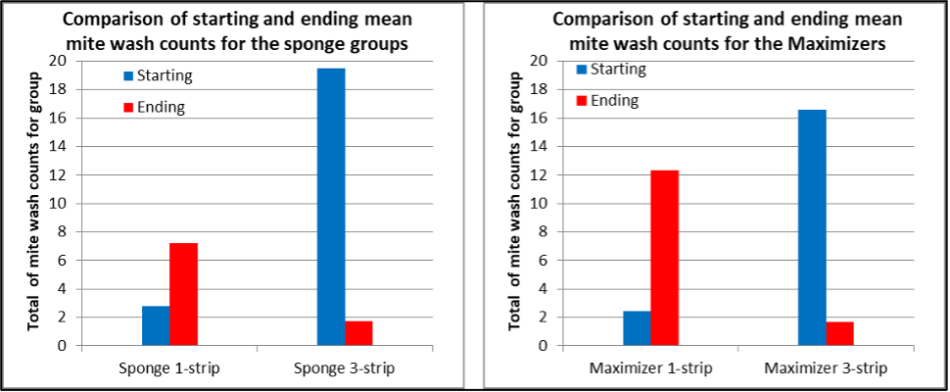
Fig. 9 Note that the 1-strip groups started with much lower mite levels, which went up, whereas the 3-strip groups started with much higher mite levels, which then went way down. The absolute reductions were 91% for the Sponges, 90% for the Maximizers. (n = 21 for 1-sponge, 12 for 3-sponge, 22 for 1-Maxi, 10 for 3-Maxi).
Those absolute reductions (as opposed to calculated efficacies) are pretty impressive in their own right, but don’t take into account the expected increase that could have been factored in had there been comparative data from legitimate controls.
To legitimately calculate efficacy of a treatment, the Test and Control groups would need to start at the same infestation rate. But a problem with testing varroa treatments is that with a high starting count, the Control colonies may succumb to the mite and virus load.
Unfortunately, one can’t just start the Control group with lower infestation rates than the Treatment group, since my previous data indicate that high-mite colonies tend to exhibit higher efficacy values than those for colonies with lower starting mite counts [[6]]. So I wondered whether that trend held over the range of starting mite counts for the 22 hives in the two 3-strip test groups combined together (Figure 10).
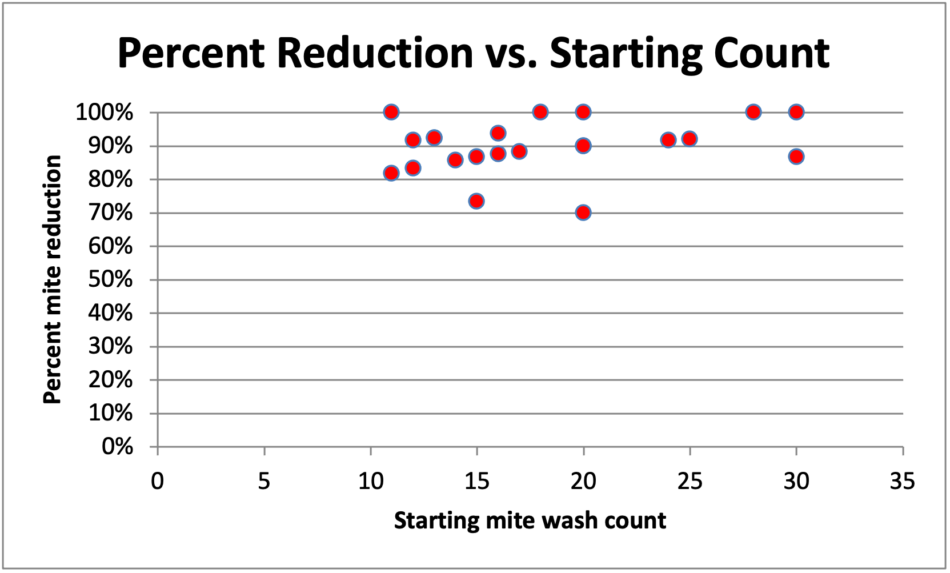
Fig. 10 Correlations between the starting counts and percent mite reduction from treatment for the 22 colonies in the two 3-strip groups combined. There was no apparent correlation.
In light of the above, and with the important caveat that any efficacy calculation for this trial cannot be considered legitimate (due to the Control group not starting at the same infestation rate), I feel that it is still informative to run the Henderson-Tilton calculation anyway, using the 1-strip applications as the “controls.” Doing so, the calculated efficacies are 97% for the Sponges, and 98% for the Maximizers!
Practical application: The 90% absolute reductions, and admittedly-questionable 97% efficacy values are what we were looking for. The much-cheaper Maximizers are looking pretty good!
TAKE HOMES:
- For full efficacy of OAE for mite control by using an absorbent matrix laid across the top bars between the brood chambers, it takes about 55 in2 of matrix holding 50 g of OA dissolved in 50 g of glycerin.
- We don’t yet know whether it helps to divide the matrix, but got good results last year with it divided into two pieces.
- Maximizer pads (Figure 11) are a lower-cost alternative to Swedish sponges, and are a bit easier to scrape off the top bars after treatment.
- We do know that strips can alternatively be draped over the top bars down between the combs with good results, but I have not done much research on this method of placement.
- My preliminary research, substantiated by recent work by Dan Aurell [[7]], indicates that Aluen CAP strips (10 g OA per strip), hung between the frames, requires more than 4 strips for equivalent efficacy in a double deep.
- Aluen CAP uses a 1:2.5 ratio of OA to glycerin; we like a 1:1 ratio; New Zealanders typically use an intermediate dilution.
- Higher-glycerin formulations (and perhaps cotton pads) release the OA more quickly, and can potentially cause short-term adverse effects.
- We’ve learned a lot so far, but need to continue our research!

Fig. 11 A dedicated paper cutter with a built-in clamp allows for quick cutting of matrices. Here are ½-sponge equivalents of Maximizer lightweight pads, which are a readily-available alternative to Swedish sponges.
Update: This is only a report on my experimental trials. I do not make any recommendations, so please do not write me for such. The Spilltech Maximizer pads above (Item # GPC100S) contain a little bit of plastic fiber, and a flame retardant (that does not appear to be harmful to the bees). We tested the lightweight version, and will test the heavyweight version this year.
To be continued…
ACKNOWLEDGEMENTS
Thanks to Brion and Alice Dunbar, my sons and crew of Golden West Bees, and to all the beekeepers who have offered suggestions and observations.
CITATIONS AND NOTES
[1] Obtaining each year a Pesticide Research Authorization from the California Department of Pesticide Regulation.
[2] Me and a few other beekeepers had a meeting with EPA’s Office of Pesticide Programs as I wrote this article in January. I’ll write about it next month.
[3] New Zealand very reasonably has determined that beekeeper use of the organic acids for mite management poses scant risk to the public or environment, allows beekeepers to use them at their own discretion. More on this next month…
[4] More on this in an upcoming article. I just returned from one of our outyards, in which 50% of the colonies have exhibited strong mite resistance this season, and required no treatment at all.
[5] Mite Control While Honey is on the Hive: Part 3 – Scientific Beekeeping
[6] https://scientificbeekeeping.com/mite-control-while-honey-is-on-the-hive-part-2/
[7] Pers. comm. and presented at the 2022 ABRC.




