The Primer Pheromones, Part 4 – Reproduction and Survival
March 30, 2011
The Primer Pheromones, Part 4—Reproduction and Survival
Randy Oliver
ScientificBeekeeping.com
First published in American Bee Journal July 2010
Queen Pheromone(s)
We humans hear the term “queen” and immediately project all the royal attributes of leadership and omnipotence upon the mother of the colony. But is this really the case? Does the queen bee really “rule” the colony, with the sterile workers acting as subordinate subjects?
The feeling that I get is that bee society is not that way at all. It appears to me that the superorganism—the colony—is the true decision maker, and that the queen functions merely as the ovary and reservoir of genetics for the colony, and that her pheromones function mainly as reports to the superorganism as to the status of the ovary. Indeed, Seeley (1995) describes “messenger bees that pick up the queen’s pheromones and then travel about the broodnest actively dispersing this olfactory indicator of the queen’s presence.”
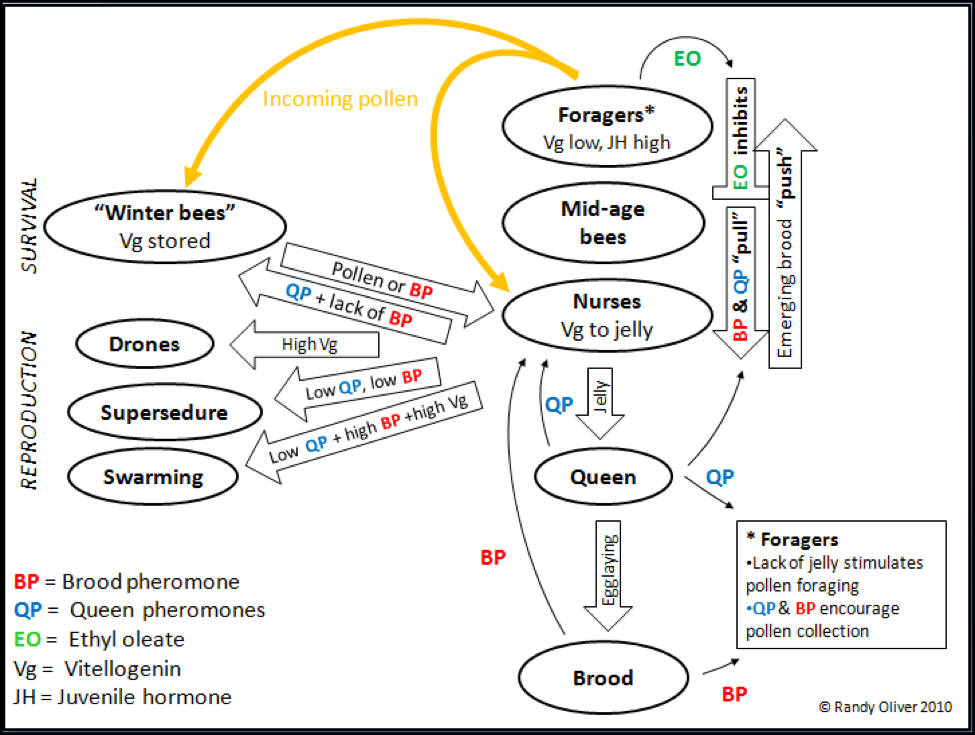
As long as the “ovary” is properly mated and functioning well, the colony is content—the presence of both brood pheromone and queen pheromone suppresses ovary development in the workers. However, should the “healthy ovary” signals falter or disappear (Fig. 1), the colony will immediately swing into action to replace the queen/ovary without hesitation or sentiment, via supersedure, or the initiation of emergency queen cells.
I’m using the term “queen pheromone” (QP) generically, since the queen produces a bouquet of pheromonal components from different glands, with multiple functions. Current terminology has moved from “queen mandibular pheromone” to “queen retinue pheromone,” since not all components necessary for retinue formation are produced by her mandibular glands. So far, nine components of the full retinue pheromone have been named, but it is clear that there are still others yet to be identified. This brings up a fascinating aspect of QP—there is a genetic component involved in worker recognition of the individual components. Some strains of bees do not recognize the queen if certain components are not present in the right amount!
Practical app: This finding makes me wonder if that is why it is difficult to introduce queens of some strains into unrelated colonies, and whether this might be related to the substantial amount of rapid supersedures sometimes observed after introducing purchased queens. If you are introducing queens of a different stock than the recipient colony, the workers simply may not recognize her pheromonal signals as being “right”!
Also of interest is that the queen apparently signals to the colony as to how well she was mated—queens that are inseminated by multiple drones produce QP that is more attractive to workers than that produced by queens mated to a single drone (Richard 2007). Kocher (2009) found “that the queen pheromone blend is modulated by the reproductive status of the queens, and workers can detect these subtle differences and are more responsive to queens with higher reproductive potential.” The quality of the QP produced by any individual queen may have considerable impact upon a colony, regardless of her genetics—Grozinger (2003) found that QP affected the expression of several hundred genes in workers!
Practical app: Poorly-mated queens (such as those forced to take mating flights during cool or rainy weather) may be quickly superseded. A well-mated queen, on the other hand, functions as a pheromonal “cheerleader” to energize and invigorate the colony, and to boost morale.
Colony Reproduction
There are two ways for a colony to disseminate its genetics (and epigenetics—we’ll get to this later) into future generations: by either the production of drones, or by division of the colony by swarming (an unusual process, in that the “parent” leaves, and the “offspring” stays behind). As you can see in Figure 1, the primer pheromones, pollen income, and vitellogenin are all involved in the “decision making” of colony reproduction.
Drone production
Compared to the risky and expensive enterprise of throwing off a swarm, the creation of expendable drones is a cheap gamble for a colony at getting a portion of its genes into the next generation. Even though any particular drone has little chance of actually mating, the colony’s investment in that drone is minimal. But by producing thousands of drones, the colony has a good chance at disseminating its genes when virgin queens from other colonies fly out to mate.
Each drone takes a certain amount of colony resources to produce and feed, so the hive carefully regulates the amount and timing of drone production. The mechanisms for such regulation are not completely understood (reviewed recently by Boes 2010). Since the only purpose of drones is to mate with flying queens (generally from other colonies), it is obvious that the best time to rear drones is when they actually have a chance at “getting lucky”—during swarming season, when the air is full of lusty young virgins. At other times of the year, the only value of maintaining a population of drones is that one may have a chance encounter with a supersedure or emergency queen.
Drones are produced, as a rule, only once a colony has reached substantial size, and then only when there is abundant pollen coming into the hive, the presence of which unleashes a cascade of pheromonal signals, resulting in a vitellogenin-rich population of nurse bees awash in jelly, and thus kicking off feedback loops of queen and brood pheromone production. When colony nutrition is poor, the freeloading drones are summarily evicted.
Practical apps: the presence of drone brood is a great indicator that a colony is in good nutritional shape; conversely, their eviction might suggest the need for supplemental feeding. Steve Taber demonstrated that one could get colonies to produce drones even during winter by feeding them pollen patties. Colonies will utilize introduced drone comb to rear more drones than they would without such added comb, but at an energetic cost to the colony (Seeley 2002).
Don’t worry about swarming at least until after a colony has produced drones—as far as I can tell, a colony invariably initiates drone production prior to building swarm cells. This makes evolutionary sense, since in bad times any rare surviving colonies would want to ensure that there were some drones present to inseminate the new queen (surprisingly, apparently no one has tested to see whether the presence of drones is essential for swarming).
Practical Tip: During pollen dearths, colonies stop rearing drones. At that time, varroa are forced into worker brood (this generally occurs during August and September). The combination of nutritional stress and high varroa infestation often leads to virus epidemics in the workers that can spell death for the colony (Sumpter and Martin 2004). Thus, this is the most critical time to monitor mite levels.
Figure 1, yet again. Note how critical the levels of queen pheromone (QP) and brood pheromone (BP) are in nearly every aspect of worker division of labor, “aging,” colony reproduction, and the transition to winter survival mode.
Swarming
Swarm production, as opposed to drone rearing, is a risky large-scale investment, with limited potential of founding a successful new colony (most swarms in nature do not survive the winter). The tradeoff is that a swarm has a much better chance at passing on its genes (all of a colony’s genetics are carried in a prime swarm –leaving a related daughter in the hive to carry on, with the infusion of genes from drones of other successful nearby colonies).
Swarming behavior is complex. It is strongly seasonal, but appears to be more directly due to colony population size (dilution of QP), the nurse to brood ratio (Vg and BP), pollen availability and the resultant abundance of jelly and Vg (Pridal and Sustek 2000), and age of the queen (QP). (I’m curious as to the ratio of open to sealed brood—in an expanding colony, there is a great deal of larvae secreting BP, but in a pre-swarming colony, most of the brood is sealed).
Lenskya (1981) observed that in crowded hives, the queen avoided the lower edges of the brood combs, and found that by applying a combination of QP and queen “footprint” pheromone to the lower edges of combs, that he could inhibit the bees from building queen cells there.
Because of the substantial economic significance of swarming to the beekeeper, management for swarm prevention has been vigorously debated, but I’ll leave that discussion for another article!
Some exceptionally large swarm cells from a strong colony that has been enjoying an abundance of forage. The queen, shortly before she is ready to emerge as an adult, emits a pheromonal signal for the workers to remove the cell wax from the tip of her silk cocoon (cell at left). Photo by the author.
Practical app: In short, keep these facts in mind: a colony tends to swarm if it has an aging queen, is in the best nutritional shape, has an abundance of stores (has filled the combs), is crowding the cavity with bees, and the queen has filled every available cell with brood. You can minimize swarming by manipulating the hive to change any of these factors during the relatively short seasonal swarm window.
Survival Mode
The colony will go into survival mode during periods of nutritional duress. The pheromonal trigger for the production of long-lived “winter” or diutinus bees appears to be the presence of QP concurrent with the absence of BP, causing bees that would normally be nurses to store Vg in their fat bodies (as opposed to converting it to jelly) in order to wait out the dearth.
Other Factors
I would be remiss not to mention that there is more to the allocation of the workforce than just the primer pheromones in Figure 1.
Worker Patrolling
The above model makes for a neat and tidy description of the colony as a factory in which all workers function as robotic automatons, devoid of thought or individual initiative. However, in reality the picture is a bit more complex, and it appears that individual bees engage in behaviors that appear to include observation, decision making, and leadership.
For example, how do individual bees “decide” whether and where they should be doing any of the other myriad jobs (beside nursing and foraging) involved in running the bee economy? And how do they make decisions such as where to build comb, and whether it should be worker or drone cell size?
Pratt (1988) found, by a series of clever experiments, that comb builders needed to make direct contact with existing drone comb (occupied or not) in order to determine whether more drone comb was needed (it’s still not clear exactly how the comb builders then manage to work cooperatively).
Another question is how returning pollen foragers evaluate the pollen needs of the hive. Dreller and Tarpy (2000) found that pollen foragers appear to “individually evaluate the [in hive] pollen to brood to empty cells ratio in order to adjust their foraging effort according to the colony’s need.”
The above behaviors are examples of jobs largely undertaken by mid-aged bees. These bees form a large generic task force of workers who have given up nursing duties, but have not yet graduated to foraging. Dr. Brian Johnson (2008, 2009) has written extensively about their importance in a colony. Bees of this “age” spend a great deal of time patrolling the hive and evaluating the colony’s needs, and then jump into any needed task. They respond to both “cues” (temperature, excessive nectar, debris or comb damage), and “signals” (such as the tremble dance, the stop signal, and the shaking signal given to them by the foragers). Johnson describes how the colony reaches a “dynamic equilibrium” through the process of mid-aged bees spending up to a quarter of their time randomly patrolling throughout the hive (often visiting every part of the nest), and then continually starting, quitting, and switching various tasks as needed. This process may not seem efficient to our eyes, but it appears to work pretty well in the bee colony at getting the various jobs done!
Individual Bee Leadership
One thing that I find intriguing is the effect of individual worker bee leadership. For example, a few “leader” bees take charge of the initiation of swarm departure. Rittschof and Seeley (2008) found that scout bees with a new nest site in mind jump start a resting swarm of bees into warming up their wing muscles, inspect them to see when they’re ready, and then give them the signal to take off! The authors explain:
It turns out that the scout bees from the chosen nest site are responsible for producing both the piping signal to prime a swarm for take-off and the buzz-run signal to trigger the take-off. We suggest that these bees produce the signal that triggers take-off because they travel throughout the swarm cluster while piping and so are able to sense when the entire swarm is hot enough to take flight. The mechanisms mediating take-offs by honeybee swarms appear to present us with a rare instance where an action of a large social insect colony is controlled by a small set of individuals that actively monitor the global state of their colony and produce a signal triggering the colony’s action in a timely way.
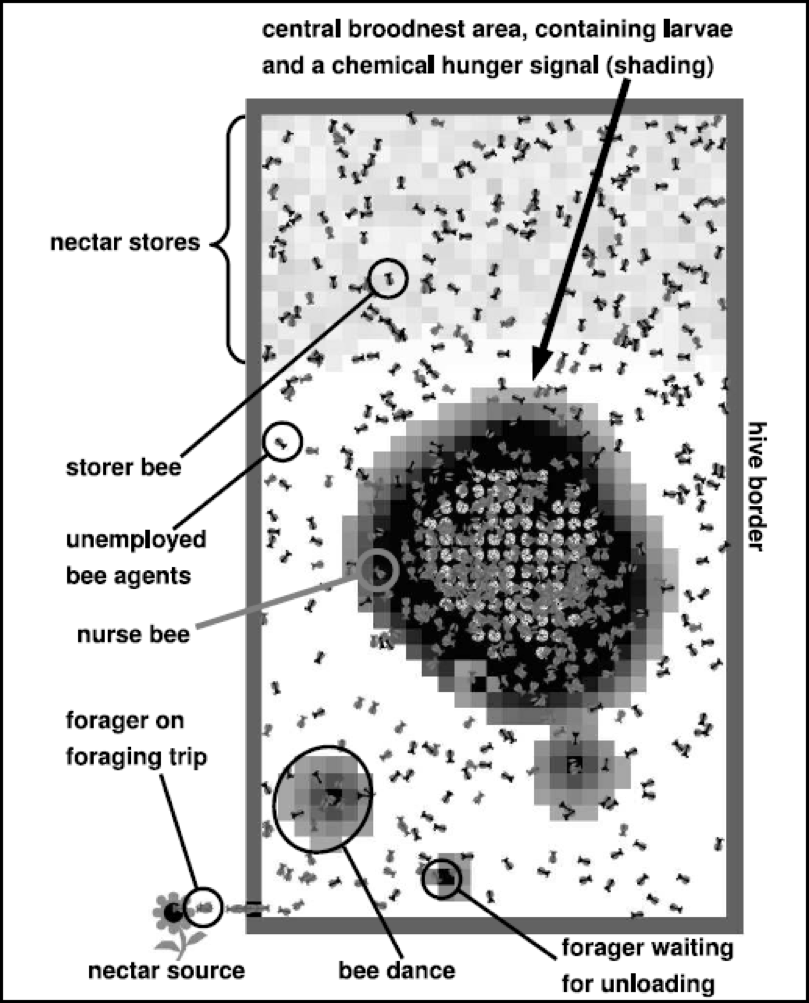
As impressive as the behavior of swarm “commanders” is, here’s something even more astounding. We’re all familiar with the famous “dance language” of honey bees, which is used to communicate the location of favorable food or water sources. This form of communication is hard wired into the bee brain, and requires no thought. However, recent research by Nieh (2010) discovered that if a forager experiences danger at a certain site (such as the smell of alarm pheromone, or the biting of its legs by predators), it may return to the hive and “stop” the dance of another forager that is directing bees back to that particular location! The alarmed bee repeatedly generates a vibrating “stop signal” and butts her head against the waggle dancer’s, causing the recipient dancer to stop dancing and recruiting. Such a remarkable warning communication sure appears to indicate an unexpected level of intelligence in an insect. However, before you go putting a “My bees are smarter than your honor student” bumper sticker on your truck, be forewarned that some dang scientist will likely figure out that it is merely an innate behavior (Figure 2)!
Figure 2. Typical screenshot of a multi-agent computer simulation by Drs. Thomas Schmickl and Karl Crailsheim (2008) that models colony division of labor. They found that live-colony experiments involving brood manipulation, food-deprivation, and colony size could be explained by the mechanisms implemented into the model. Models such as this help us to test our understanding of colony dynamics and behaviors. Courtesy Karl Crailsheim.
Genetics and Temperature
Of course, all the above behaviors vary greatly depending upon the genetics of each bee strain and the subfamily of sisters in each hive. The environment both outside and in the hive also has an effect. Dr. Jürgen Tautz (2003) found that “the temperature at which pupae are raised will influence their behavioral performance as adults and may determine the tasks they carry out best inside and outside the hive.”
Figure 3. A student model of brood comb heating based upon the thermodynamic model of Humphrey and Dykes (2008). The dark red cells represent heat-generating workers. Slight differences in temperature during brood development can affect later bee behavior. Modified from Thermal Conduction in Bee Hives (2009). Courtesy Martin Bracke.
Synthetic Pheromones
There are currently a few synthetic bee pheromones on the market, with the Canadian company Contech Enterprises Inc. (formerly Phero Tech) offering both queen mandibular pheromone (Pseudoqueen—formerly BeeBoost) and brood pheromone (SuperBoost). I have spent considerable time in discussion with principal scientists from the company, and find them to be quite open, earnest, and helpful. Their products offer new possibilities in hive management.
Synthetic Queen Pheromone
There are often times when a beekeeper simply wishes that he or she had an extra queen on hand. In many of those instances, a Pseudoqueen strip could be used as a temporarily stand in for the real thing.
Practical applications:
- Use to keep queenless packages “happy”
- Increase the acceptance of queen cells within a colony
- Improve queen mating success and mating nuc stability
- Capture straggler bees, mop up after swarm removal, or to trap stray bees at gas stations, etc. (add a bit of orientation pheromone or lemongrass oil to enhance the attractiveness)
- Increase pollen foraging in young, growing colonies (but apparently not as much in established colonies–Higo et al 1992)
- Perhaps for decreasing the propensity to swarm?
- Make queenless “disposable pollination units”
- Temporary queen replacement in a variety of situations
- Here’s an interesting tip from my friends in Australia to avoid Small Hive Beetle problems in mating nucs. They say that SHB explodes when you pull a queen and put in a new cell. However, if you put 1/3 of a Pseudoqueen strip in, the beetle doesn’t cause a problem, yet the new queens still mate out fine.
- I’m sure that ingenious beekeepers will come up with more uses!
Synthetic Brood Pheromone
This new product (SuperBoost) makes some pretty extravagant claims:
SuperBoost stimulates foraging, aids in the revitalization of overwintered colonies and increases honey production.
|
So are the Sales Department’s claims hype or could they really be true? (One company scientist is “much more comfortable” with a claim for 50% or greater honey production). From a biological aspect, refer back to Figure 1 to see how the level of BP affects nearly every aspect of hive regulation. Sagili and Pankiw (2007) tested SuperBoost in observation hives, and found that “queens in the BP treatment laid more eggs, were fed longer and were less idle compared to controls.” Extra BP appears to supercharge a colony’s appetite for food, and to keep it in “buildup mode.”
It’s one thing to use a synthetic queen pheromone strip as a temporary fake queen to keep bees happy, but something else entirely to make claims that production by our already hardworking bees could be doubled by the application of synthetic BP. Long-time beekeepers are understandably skeptical as hell about such claims, and justifiably wonder if there will be a downside later that will come back to bite them. Contech is very much aware of this concern, and has been tracking the health and survival of colonies after long-term use of the product. To date, this concern appears to be unfounded—the colonies appear to thrive.
I’ve been over the data from Contech’s cited field trials (e.g., Pankiw 2008), plus yet unpublished data from more recent trials provided me by Chief Scientific Officer Dr. John Borden. I must say, it does appear that SuperBoost can supercharge colonies, at least at certain times of the year. Pankiw’s trial was during late winter in Texas; a different unpublished trial in British Columbia ran during spring and summer. Certainly, more trials and practical use by commercial beekeepers will be needed to see under which conditions the product works best, the most effective timing, and for what purposes it will be cost effective.
Of course, there is no free lunch, and it appears that in this case you will need to provide the bees lunch—in the form of pollen supplement to get full benefit of the added pheromone. This makes perfect sense—if additional brood pheromone stimulates the bees to produce more jelly, then they are going to need supplemental protein to produce it.
Practical note: The addition of BP increases the nurses’ appetite for pollen—be sure to provide supplemental protein if necessary.
So would introducing additional brood pheromone be a relatively natural way of optimizing colony buildup, or would it be akin to putting your hive on steroids, with unintended consequences? I spoke with Mike Campbell of Campbell’s Gold, who ran the Canadian trials last year. He is quite pleased with SuperBoost, and continues to use it to initiate early spring buildup, to induce colonies to take feed and to build up during dearth, and to reinvigorate fall colonies prior to wintering. He hasn’t noticed any downside.
Practical questions:
- Could synthetic brood pheromone be used in queen cell builders to encourage jelly production?
- Could it be used to increase the harvest from pollen traps?
- Smedal’s (2009) research suggests that exposure to brood pheromone inhibits the ability of young bees to become long-lived “winter bees,” so one might wish to be cautious in fall if you’re in a cold-winter area. Note, however, that based upon favorable anecdotal observations, Contech plans to run fall trials.
- Could SuperBoost be used midwinter prior to almond pollination to encourage early buildup? I’ll publish results of a small test that I ran!
I’d like to add an important comment from Dr. Cameron Lait of Contech: synthetic BP treatment should never be thought of as a magic cure for weak or diseased colonies, or those for those with poor queens. In order to realize its potential, you should only apply it to healthy, well-fed colonies, with strong queens. The colony must also be large enough—about 10,000 bees (a 3-lb package, or 5-frame nuc).
Use in Commercial Pollination
SuperBoost Revolution in Pollination
|
The above claims from the sell sheet targeted at growers may sound a bit implausible if you do the math, especially when you consider that BP encourages workers remain in the brood nest. However, in an early experiment, Pankiw (1998) found that brood pheromone stimulated pollen foraging by workers. She later (2004) found that synthetic brood pheromone showed promise for increasing bee pollination efficiency in the target crop (cucumber and zucchini). However, for the #1 bee-pollinated crop, data from Dr. Frank Eischen (in prep) did not find a substantial pollination effect from using synthetic brood pheromone in colonies during almond pollination (since that study, the company has made significant improvements to the design and storage of the device, which could affect the efficacy). The high cost for pollination of almonds will likely generate more trials.
I’ve seen additional unpublished data from carrot pollination that looks like SuperBoost treatment substantially increased bee visitation. There are other difficult-to-pollinate crops, such as seed onions, kiwi, certain cherries, and in the production of hybrid seed, in which any enhancement of bee visitation would be of value. It remains to be determined in which crops synthetic pheromonal stimulation of rented colonies might be cost effective.
This series of articles on the primer pheromones grew far beyond my initial expectations. I hope that you found the subject to be as fascinating as I did! I truly feel that a good understanding of how the primer pheromones modulate colony dynamics can help each of us to be better practical beekeepers.
References
Boes (nee Wharton), KE (2010) Honeybee colony drone production and maintenance in accordance with environmental factors: an interplay of queen and worker decisions. Insect. Soc. 57(1): 1-9.
Bracke, M, et al (2009) Thermal Conduction in Bee Hives, http://www.im.pwr.wroc.pl/~hugo/HSC/imprezy/ ModellingWeek/reports/Report_project02.pdf
Dreller C, Tarpy DR. (2000) Perception of the pollen need by foragers in a honeybee colony. Anim Behav. 59(1):91-96.
Grozinger, CM, NM Sharabash, CW Whitfield, and GE Robinson (2003) Pheromone-mediated gene expression in the honey bee brain. PNAS 100 (2): 14519–14525.
Higo, HA, SJ Colley, ML Winston, and KN Slessor (1992) Effects of honey bee (Apis mellifera) queen mandibular gland pheromone on foraging and brood rearing. The Canadian Entomologist 124: 409-418.
Humphrey, JAC and ES Dykes (2008) Thermal energy conduction in a honey bee comb due to cell-heating bees. Journal of Theoretical Biology 250: 194–208.
Johnson, BR (2008) Global information sampling in the honey bee. Naturwissenschaften 95:523–530.
Johnson, BR (2009) A self-organizing model for task allocation via frequent task quitting and random walks in the honeybee. The American Naturalist 174(4): 537-547.
Keeling, C. I., Slessor, K. N., Higo, H. A. & Winston, M. L. (2003) New components of the honey bee (Apis mellifera L.) queen retinue pheromone. Proc. Natl. Acad. Sci. USA 100, 4486–4491.
Kocher (nee Ayroles), SD (2009) Molecular and physiological mechanisms underlying chemical communication in the honey bee, Apis mellifera. http://www.lib.ncsu.edu/theses/available/etd-08112009-235911/
Kocher, D, F-J. Richard, DR. Tarpy, and CM Grozinger (2009) Queen reproductive state modulates pheromone production and queen-worker interactions in honeybees. Behav Ecol 20 (5) 1007-1014.
Lenskya, Y and Y Slabezkia (1981) The inhibiting effect of the queen bee (Apis mellifera L.) foot-print pheromone on the construction of swarming queen cups. Journal of Insect Physiology 27(5): 313-323.
Nieh, J. (2010) A Negative Feedback Signal That Is Triggered by Peril Curbs Honey Bee Recruitment, Current Biology 20:1-6.
Pankiw, T, RE Page, and MK Fondrik (1998) Brood pheromone stimulates pollen foraging in honey bees (Apis mellifera). Behavioral Ecology and Sociobiology 44: 193-198.
Pankiw T. (2004) Brood pheromone regulates foraging activity of honey bees (Hymenoptera: Apidae). J Econ Entomol. 97(3):748-51.
Pankiw, T, RR Sagili, and BN Metz (2008) Brood pheromone effects on colony protein supplement consumption and growth in the honey bee (Hymenoptera: Apidae) in a subtropical winter climate. J. Econ. Entomol. 101(6): 1749-1755.
Pratt, SC (1998) Decentralized control of drone comb construction in honey bee colonies. Behav Ecol Sociobiol 42: 193-205.
Pridal, A. and Sustek, D. (2000) Development of hypopharyngeal glands in honeybee workers during growth and swarming fever of their colonies. Pszczelnicze Zeszyty Naukowe, 44(2), 25-34.
Richard F-J, Tarpy DR, Grozinger CM (2007) Effects of insemination quantity on honey bee queen physiology. PLoS ONE 2(10): e980.
Rittschof, CC and TD Seeley (2008)The buzz-run: how honeybees signal ‘Time to go!’ Animal Behavior 75: 189-197.
Sagili, RR and T Pankiw (2007) Brood pheromone regulation of queen egg-laying in the honey bee (Apis mellifera L.). Presented to the Entomological Society of America.
Schmickl, T and K Crailsheim (2008) Analysing honeybees’ division of labour in broodcare by a multi-agent model. Artificial Life XI: 529-536.
Seeley TD (1995) The wisdom of the hive: the social physiology of honey bee colonies. Harvard University Press, Cambridge.
Seeley, TD (2002) The effect of drone comb on a honey bee colony’s production of honey. Apidologie 33: 75–86.
Smedal, B, M Brynem, CD Kreibich and G. V. Amdam (2009) Brood pheromone suppresses physiology of extreme longevity in honeybees (Apis mellifera). The Journal of Experimental Biology 212: 3795-3801.
Sumpter, D. J. T. & Martin, S. J. (2004) The dynamics of virus epidemics in varroa-infested honey bee colonies. Journal of Animal Ecology 73:51-63 www.lasi.group.shef.ac.uk/pdf/Martin%2064.pdf
Tautz, J, S Maier, C Groh, W Rossler, and A Brockmann (2003) Behavioural performance in adult honey bees is influenced by the temperature experienced during their pupal development. Proceedings of the National Academy of Science 100(12): 7343–7347.
Wright, W (various) www.beesource.com/point-of-view/walt-wright/