An Experiment to Improve Pollen Sub: Part 2
Beekeeper-Funded Research
An Experiment to Improve Pollen Sub: Part 2
Randy Oliver
ScientificBeekeeping.com
First published in ABJ May 2020
In my last article I laid out the reason that I ran this experiment, and my two suspects for being the limiting nutrient in the artificial diet – the lack of an important sterol and/or a deficiency of zinc.
objectives of the experiment
hypothesis to test
That zinc and/or 24-methylenecholesterol (24-mCh) are the limiting nutrients in the commercial pollen sub that I used, and that by increasing their amounts in a supplemented Test batch, the performance of the diet, as measured by the amount of colony growth, would improve.
expected benefit
Since protein is typically the most expensive component in a diet, a balanced formulation (not limited by zinc or sterol content) would allow more complete utilization of the total protein, and thus better cost efficiency of that diet.
experimental design
We utilized the extended period of pollen dearth in the Sierra Foothills — during which colonies typically decline in strength — to compare the amount of colony growth resulting from the feeding of four different diets:
- Positive Control: a benchmark “optimal” diet consisting of blended bee-collected pollens from three sources, mixed with Drivert sugar and Pro-Sweet syrup to match the protein and sugar content of the two pollen sub patties.
- Negative Control (no protein): a benchmark protein-free diet consisting of Drivert sugar and Pro-Sweet syrup to match the sugar content alone of the Control and Test patties.
- Control: Off-the-shelf Ultra Bee Bulk Soft patty.
- Test: Ultra Bee Bulk Soft patty supplemented with zinc and 24-mCh from borage oil replacing the canola oil in the Control patty.
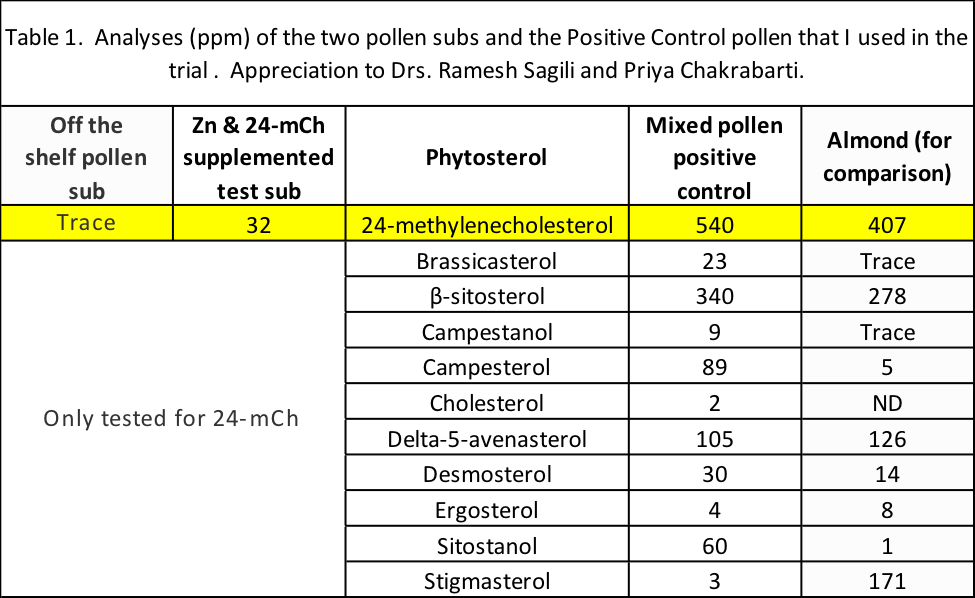
Summary: So of the four diets to be tested, two – the Positive Control natural pollen, and the Test (Supplemented) formulation — would contain appreciable quantities of 24mCh (although there was much more in the natural pollen patties). On the other hand, the Control (off-the-shelf) and Negative Control (sugar) patties would contain only a trace or none, respectively (Table 1).
The colonies and environmental conditions
We started with equalized 5-frame nucs with young queens, varroa controlled, and attempted to grow them to maximum size during the highly unfavorable summer/fall/winter dearth period in the Sierra Foothills. .
Practical application: Running a meaningful test of artificial diet requires an environment in which natural pollen is lacking, since as was evident in my 2013 trial and another study I performed [[1] ], there is scant or no benefit from feeding pollen sub when there is a natural pollen flow on.
We would equalize the colonies to start, and then grade them for cluster strength at three additional time points, with the final grading taking place shortly before the financially-important almond pollination season. The trial would thus mimic the situation and conditions faced by inland California beekeepers attempting to grow late nucs for almond pollination.
Materials and methods
In mid-July we had a bunch of late nucs in a yard, ready to be moved to a better location. Instead, we distributed spaced pallets and transferred the nucs to singles on the pallets (Figs. 1-3).
Figure 1. The experimental yard after transfer. Pallets well-spaced and cocked, all four entrances on each pallet facing in different directions, in order to minimize drift. At this point, they were ready to equalize.
On 6 August we began equalizing the colonies to 5 frames covered with bees, replacing any colonies with poor queens or brood.
Figure 2. In order to equalize the colonies, we only install 5 frames at first, so that we could quickly determine cluster size by eyeballing the bee coverage of the exposed outer frames. I find this technique works well for equalizing cluster sizes. The blue towels contained oxalic acid and glycerin for mite management; this application method is not yet approved by EPA [[2]].
Figure 3. We used a sieve box to shake bees from stronger colonies into weaker ones, over the course of three days. At the final equalization, each colony contained three frames with brood, with the outer comb sides each ¾ covered with bees, making sure that the four hives on each pallet were very closely equalized (for a good randomized block design).
At this point we filled the hives with another five frames of drawn comb, numbered the hives, and randomly assigned their treatment group, evenly distributed to each pallet. There were 13 each of Negative and Positive Controls, and 26 each of the two pollen sub types. Now we were ready to start feeding them.
Preparation of the test patties
I’d requested Mann Lake to provide the Test and Control pollen sub formulations weighed into 1-lb patties between waxed paper. I obtained analyses and recipes for the patties, from which I could calculate the protein content (67 g/patty), as well as the amount of added sugar (234 g/patty). I then formulated the Positive and Negative Control patties on-site with matching protein and/or sugar contents.
For the Positive Control patties, I used trapped cleaned pollen from three sources — generously donated by two beekeepers who sell pollen commercially — Jeff Becker (California Bee Pollen) and Derrick and Melissa Maness (Colorado Mountain Honey). I homogenized it together, and mixed in measured amounts of Drivert and Pro-Sweet sugar (Figs. 4 & 5), which resulted in a patty weighing a little better than a pound and a half (717 g), compared to 1 pound (454g) for the Test and Control patties. The Negative Control patties were smaller, weighing a half pound plus (247 g).
Figure 4. We carefully weighed the ingredients of the Pollen Positive Control and Sugar Negative Control patties and mixed them by hand.
Figure 5. Here Brooke and I are weighing Pollen Control patties to the gram. We mixed these patties fresh for each feeding.
Abbreviated Experimental Log
9 August – We randomly assigned treatments to the four hives on each pallet, and applied the first round of patties (Fig. 6) and a gallon of Pro-Sweet syrup mixed 3:1 with water (61% sugar), with a half cup of bleach per 50 gal to retard fermentation. We fed ½ gal of syrup again roughly once a week to keep the small colonies stimulated.
Figure 6. Brooke feeding the colonies (empty boxes are above the singles to insulate them from solar radiation). Patties got laid out by group assignment and double-checked by a second person prior to feeding. The hives were set up in a randomized block design, four hives per pallet, entrances randomly assigned to face different directions to avoid drifting. All patties contained the same amount of added sugar, and were matched for protein content, other than the protein-free Negative Control. Note the dry environment, with provided only occasional small amounts of natural pollen.
We continued to feed syrup regularly to stimulate colony growth. Unexpectedly, there was a slight amount of natural pollen coming in from the landscape from time to time.
23 August — Most of the colonies had consumed all or most of their first patty, so we added another round. There was no noticeable difference in consumption between the Test and Bulk patties, so the borage oil did not appear to have an effect upon consumption in either direction. I spot checked several hives—all were rearing brood in earnest. There was some, but not much natural pollen in the combs.
11 September — We added a 2nd brood chamber (containing 4 frames of extracted drawn comb, 5 frames of foundation), the third round of patties, plus 2 Apivar strips (Fig. 7), and continued regular syrup feeding. There was very little natural beebread in the combs.
Figure 7. Good to the last smidgen—Bulk Patty and Apivar strips. Surprisingly, the pollen sub patties were being consumed at roughly the same rate as the natural pollen Positive Controls.
21 September — Patty feeding #4. Colonies OK, but many not yet building substantially. No brood at all in the upper drawn combs of 10+ hives that I checked. Checked for jelly around 2nd-instar larvae—abundant jelly in all groups other than Sugar Control. Although Sugar Controls had good-looking brood, it appeared that they may be cutting back on jelly feeding. In general, there was less sealed brood than I’d hoped to see—perhaps due to the warm, dry weather. We’ve got about two full brood cycles left for the colonies to grow before November slowdown. Fed ½ gal syrup on Thurs, another on Monday, in order to sustain an artificial nectar flow.
26 September — Checked a Sugar Control: a fair amount of natural beebread on one frame.
4 October — Patty feeding #5. It occurred to me at this time that the supplemented Test patties might be lacking in omega-3 fatty acids, so I mixed up canola oil with Drivert sugar and started applying a scoop each time we fed a patty, to both the Control and Test groups (Fig. 8).
Figure 8. Belatedly realizing that the borage oil may not be providing enough omega-3 fatty acids to the Test patties, we started applying a scoop of canola oil patty to both of the pollen sub groups along with their patty.
16 October — Patty feeding #6, along with a scoop of canola oil patty. Big Bummer—yellow pollen in the hives. Yet patty consumption was very good. The bees have filled the 4 drawn combs in the upper boxes with honey—no brood up there. But very little drawing of any foundation. Frustratingly, colony variability is huge; and no apparent correlation between colony strength and patty consumption, nor do I observe any clear correlation between buildup and patty type.
30 October —Time point #1 grading (end of season). Patty feeding #7. Started grading at 7:00 am, cold and dark, finished by 8:00, clusters still tight.
30 November —Removed and weighed any uneaten patties (no consistent differences). Spot checked for brood. Checked one hive each for protein-fed groups—all had a fair amount of open brood, plus a patch or two of sealed brood. Checked 4 Negative Control hives—not all had sealed brood; only one had a small amount of open brood. Conclusions—feeding of protein extended the duration of broodrearing, but at a fairly low level.
4 December — Time point #2 grading (winter minimum cluster size).
27 December —Started feeding patties and syrup again (patty feeding #9
14 January — Last patty feeding (#10). Some alder pollen coming in between breaks in the rain, but rain and cool weather predicted. Large within-group variation of consumption—some smaller clusters had consumed every bit of their patty, whereas some larger clusters still had at least half remaining.
28 January — Time point #3 grading (pre-almond cluster size). Graded midday with a little flight going on (in order to compare to normal almond grading) (Fig. 9). Large amounts of alder pollen coming in and stored in the combs. Even the Sugar Control hives typically had 2-3 frames well-covered with mostly sealed brood; the stronger hives in the other test groups had even more brood. When I checked the sealed brood for age, it was clear that the adult bee populations were from brood reared prior to alder pollen coming in. Almond bloom is still at least two weeks away, so we expect that the clusters will be substantially larger at grading time, due to the emergence of the frames of sealed brood.
Fig. 9. By the end of January, we needed to grade the colonies for strength prior to any workers emerging from brood that was reared once alder pollen supplied nutrition other than from the test diets. It was still three weeks from when the hives would be graded for strength in the almond orchards.
Three very weak hives had water pooled on the bottom boards, so were excluded from analysis. We took mite washes from 8 hives to confirm varroa control by the Apivar strips—didn’t find a single mite. The trial was over.
summary
The trial ran from 9 August through 28 January (172 days), during which minimal natural pollen or nectar were available, at an elevation that seldom sees a winter frost.
Each colony received 10 feedings of patty, for a total of 10 lbs of pollen sub, just under that amount of natural pollen (with additional sugar added), or sugar patty alone. Each colony received 17 half-gallon feedings of 61% sugar syrup (9½ gallons in total).
As we’ve found in previous years during our summer dearth, it’s difficult to build nucs started after June up into strong colonies for winter. Even with considerable nonstop feeding, they just don’t grow at the same rate as nucs started in the springtime. But they clearly grew enough to see the benefit of feeding supplemental protein, whether of natural pollen or pollen sub.
Results
It’s easy to convince oneself you’ve got a great idea. But in this case, hard data did not support my hypothesis that supplementing pollen sub with zinc and 24-mCh would improve its performance (although I suspect that zinc is still a good idea). The results are graphically (and disappointingly) displayed in Fig. x.
Figure x. There was a clear difference in performance between the colonies fed natural pollen and the sugar control (p = 0.002), but not so much between natural pollen and the pollen subs. I arbitrarily excluded any “problem” colonies with disease or other unidentified issues. Of possible interest is that there were no “problem” colonies in either the Pollen Control or Sugar Control groups, despite one group being stronger on average, and the other weaker than the pollen sub groups.
Practical application: In the above graph, the down-and-up changes in cluster size may largely be an artifact of temperature rather than population. Note how close the end-of-January sizes reflect the end-of-October gradings.
Discussion
This trial was intentionally run under poor conditions for colony growth. In springtime, a 5-frame nuc in this same location can easily grow on natural forage to a 20-frame colony in two months. Not so for a nuc started in July, which typically won’t grow at all. I had hoped that by providing natural pollen ad libitum, along with abundant sugar syrup, that I might be able to stimulate the natural pollen positive control group to at least build up to 15 frames of bees. That didn’t happen – by the end of October, only two of twelve had reached 10 frames in strength, with an overall mean cluster size for the group of only 8.5 frames.
The results indicate that:
- Colonies don’t grow well in my environment in late summer, even if they are abundantly fed with natural pollen and sugar syrup.
- Honey bees appear to be able to convert other sterols into 24-mCh.
- There was no apparent benefit from the high level of 24-mCh in the natural pollen Positive Control patties, compared to the mediocre level in the borage oil Test patties.
- Canola oil apparently works well as a lipid and sterol source in pollen sub, even though it is deficient in 24-mCh.
- A good pollen sub can perform, on a weight to weight basis, better than natural pollen. But natural pollen appeared to hold a slight lead on a per gram protein basis (Table 2).
- An unanswered question is why the excluded sickly colonies appeared only in the pollen sub groups – perhaps due to gut dysbiosis of some sort?
- In any case, this trial confirmed the clear economic benefit, under the conditions of this trial, of feeding pollen sub (Table 2).
| Table 2. Return on Investment from Feeding Pollen Sub | ||
| Mann Lake
Bulk Soft Patty |
Positive Control Patty of natural pollen and sugar | |
| Weight (mass) of each patty (g) | 454 | 717 |
| Amount protein per patty (g) | 67 | 67 |
| Amount of added sugar per patty (g) | 234 | 234 |
| Total patty mass fed per hive in the 10 feedings (g) | 4540 | 7170 |
| Mean amount of patty residue not consumed (g) | 352.6 | 107.5 |
| Net patty consumption (lbs) | 9.2 | 15.6 |
| Median colony strength* gain over sugar controls | 2.5 | 3 |
| Net gain in strength per pound patty consumed | 0.27 | 0.19 |
| Net gain in strength per gram protein consumed | 0.37 | 0.45 |
| Rental rate in almonds per frame of bees | $25.00 | $25.00 |
| Dollar return per pound of patty fed | $6.78 | $4.82 |
| Cost per pound for pollen sub | $1.20 | |
| Return on investment (not counting labor) | 465% | |
| * Measured as frames covered with bees for almond grading. The strength difference would likely have been even greater by bloom time. | ||
Practical application #1: My cost, with labor factored in, of feeding the sugar alone group was around $43 per nuc. On average, those nucs didn’t make 6-frame minimum grade for almonds. Adding an additional $12 for feeding pollen sub, allowed us to rent 8-framers for $200, a nice return on investment.
Practical application #2: For colonies that are already well-established in doubles, the feeding of 8-10 lbs of pollen sub during September and October (after serious varroa control in August), in our experience makes all the difference in the world for bringing premium hives to almonds (better than $200), due to that feeding resulting in the production of a generation of workers that will form a larger and better-nourished winter cluster.
A follow-up field trial
Since there are new pollen subs on the market since I ran the comparative trial in 2013 (including one of spirulina), I’m currently planning to run another large-scale comparative trial this summer, and would appreciate funding donations from those interested in seeing the results.
Acknolwedgements
I thank the donors to ScientificBeekeeping, whose generous contributions are my sole source of funding for all my research projects.
Thanks to Mann Lake Ltd. for preparing and donating the Test and Control patties, and the Pro-Sweet syrup for the trial. Thanks also for the donations of pollen from Jeff Becker at California Bee Pollen [[3]], Derrick and Melissa Maness at Colorado Mountain Honey [[4]], and Paul Limbach (Western Colorado Honey). Much appreciation to Dr. Priya Chakrabarti for the sterol analyses.
References & Notes
[1] https://scientificbeekeeping.com/when-to-feed-pollen-sub/
[2] I obtained a Pesticide Research Authorization from the California Department of Pesticide Regulation.
[3] https://www.californiabeepollen.com/ 916-396-1697
[4] https://www.coloradomountainhoney.com/