IPM 2 Fighting Varroa 2: Choosing your Troops: Breeding Mite-Fighting Bees
IPM 2 Fighting Varroa 2
Choosing your Troops
Breeding Mite-Fighting Bees
(Second in a series on Integrated Pest Management of varroa)
ScientificBeekeeping.com
Originally published in ABJ, Jan. 2007
I got tired of getting my butt kicked by varroa. My first step in getting the upper hand on the mite was to forswear the coddling of wimpy bees with synthetic chemicals. This decision cost me dearly as colonies collapsed right and left. But thanks to the genetics of selected mite-resistant queens, my colonies now look better than they have in many years, and every box is again full of healthy bees.
Update June 25, 2012 For a great webpage on the biology of varroa mite reproduction, see http://www.ars.usda.gov/services/docs.htm?docid=2744&page=14
Update 12/2009 I foreswore synthetic miticides in the year 2000. My bee stocks are not yet completely varroa resistant, but do a good enough job that I now consider varroa to be more of a nuisance than a demon. I ran 700 colonies this summer, and run my bees next to large commercial operations, so my bees are exposed to the full gamut of mites and other parasites.
Author’s note: I’d like to dedicate this article to Steve Taber, the iconoclastic researcher and bee breeder who continually challenged us to question everything we “know” about bees and beekeeping.
Introduction to mite tolerance
A beekeeper friend asked me the other day, “Why haven’t those danged researchers found us the cure for varroa yet?” I replied, “They have—Marla Spivak’s been on her soapbox for years, telling us what it is: Stop perpetuating bees that can only survive with chemical help.” We’ve all seen how the mite develops resistance to each new chemical; bees will do exactly the same to develop resistance to the mite, if we just allow selective pressure to exert its effect. Well, duh! What’s it going to take until we all realize this? Here’s the skinny from the scientific community: they’re tired of looking for “Silver Bullets.” There are a few new chemicals coming down the line, but bee scientists in general are telling us that we need to move beyond the numbskull beekeeping practice of throwing a mite bomb into our hives once a year until it’s ineffective, but rather start practicing smart beekeeping, or IPM, centered around fightin’ bees that kick mite butt with only occasional help from us.
On its natural host, Apis cerana, varroa is a minor pest. Cerana has evolved a number of defenses against the mite—notably grooming behavior, infested worker brood removal, and trapping the mite in drone brood. These defenses allow the mite to coexist in the colony, but at low levels. This is an expected mature host/parasite relationship. The European honey bee, Apis mellifera, rarely showed any such traits when first infested with varroa, but these days we’re seeing the behaviors for chewing out infested brood and grooming more commonly.
If there had been no human intervention after varroa made the jump to the European honey bee (EHB), the vast majority of colonies would have died out, but, given a large enough population, and with enough genetic variability, the few survivors would likely have repopulated some or all of the range. The process would have been encouraged by the selective pressure against severely virulent strains of mite—they would have had a hard time dispersing over honeybee-impoverished areas without human assistance (robbing being critical to the dispersal of virulent mites).

This is a colony on the verge of collapse due to the combination of varroa and Deformed Wing virus. Note the circled mite and the bees with deformed wings.
The downside to this process, however, is that agriculture would have suffered; and beekeepers would have gone out of business, what with only a fraction of a percent of colonies initially surviving the varroa onslaught (as in my operation when the mite first hit—I lost all but six colonies out of about 250). So we desperately launched into a pitched battle consisting largely of throwing chemicals at the mite. This strategy worked okay at first, but, as we’ve found, we were simply perpetuating wimpy bees, and inadvertently executing a concurrent breeding program for virulent “supermites.”
The feral population of bees, left to their own, also largely perished. But we inadvertently hampered their efforts to evolve resistance to the mite by flooding drone congregation areas with mite-susceptible drones from our managed yards, and filling hollow trees with mite-infested swarms, which would then collapse in a year or two. Nonetheless, we’re seeing feral populations starting to survive. This brings up a good point: If you’re not part of the genetic solution of breeding mite-resistant bees, then you’re part of the problem. Every time you allow drones or swarms to issue from a colony that owes its survival to a miticide application, you’re hindering the natural process of evolution toward mite-resistant bees!
Using mite resistant bees
So what is one to do? First, promote mite resistant queens, either by purchasing from a breeder/producer, or taking on the challenge of selecting and raising your own (covered below). (Mite “resistance” implies active fighting of the mite; mite “tolerance” includes viral resistance, or other tolerance mechanisms. For the sake of simplicity, I will generally use the term “mite resistant.”) There are a number of beekeepers throughout the world doing exactly this, either formally or informally, many with considerable success, in both small and large operations. There are several researchers in this country doing the tedious grunt work involved in selection, such as Steve Taber, Marla Spivak, John Harbo (retired) and Jeff Harris, Sue Cobey, Latshaw and McAdams, and Tom Rinderer to name a few, as well as dedicated private parties, such as Dan Purvis, Danny Weaver, Kirk Webster, and Joe Waggle (I know I’ve inadvertently left some of you out—these names just came to the top of my head).
Funny thing is, when I talk to the large queen producers, mite tolerance is not the main thing buyers are asking for! (The same thing happened years ago when Steve Taber developed a tracheal mite resistant line). Most breeders are selecting for mite tolerance anyway (even if inadvertently), but I’ll tell you, they are going to respond to customer demand. That said, there are few, if any, open-mated stocks that are completely “mite proof,” so producers are hesitant to promise you the moon (and you may be disappointed by those that do!). Also, many beekeepers unfortunately had poor experiences with the first generations of bees touted to be mite resistant. For example, when John Harbo first released the SMR (suppression of mite reproduction) queens, he was clear that they were not intended to be used for production stock. Despite his caveat, a number of beekeepers (myself included) used them as such, and were disappointed by their performance in the field. Through no fault of the SMR queens, some beekeepers are of the “once bitten, twice shy” mentality and are hesitant to try “resistant” stock again.
Another reason for not getting on the bandwagon is, as one of my commercial friends says: Why should I sacrifice five pounds of honey yield by buying mite-resistant stock, when I’m controlling the mite just fine with popsicle sticks dipped into the latest agricultural chemical? Their point is: there is no free lunch, i.e., the bees have their hands full—and if you give them a new job to do, they will have to cut elsewhere. A review by Steve Shepard (2006) considers such tradeoffs of mite resistance vs. honey production. However, in the Big Picture colony stress due to mite damage and miticide residues may well cost the colony more in production than behavioral resistance would. After reviewing yields by mite-resistant colonies vs. Starline bees, Spivak and Reuter (2001) conclude: “colonies bred for hygienic behavior suffer no apparent fitness costs, and perform as well, if not better, than commercial stocks not bred for hygienic behavior…” It doesn’t appear that lack of production is a necessary fault if one makes a point of breeding only from the strongest and most productive colonies.
Breeding for mite tolerance
I’m not going to explain to you here how to rear queens. But if you already know how, let me make some suggestions for selecting for mite tolerance. Essentially, you want to determine which colonies are handling varroa without help, and breed only from them. John Harbo and Jeff Harris collected “survivor” queens from apiaries that had collapsed from varroa, and bred from them, selecting for the most mite resistant. Due to an initial misinterpretation of their mode of action in fighting varroa, they labeled them SMR, but now refer to them as VSH (varroa-sensitive hygiene) bees. It appears to me that VSH behavior is one of the traits exhibited by all current mite-resistant stocks of bees. An added advantage of hygienic bees is that they clean up other brood diseases, such as AFB and chalkbrood on their own!
Varroa Sensitive Hygiene (VSH)
VSH bees have a simple tactic for fighting varroa—they simply chew the buggers out of the capped cell when they try to reproduce (Figure 1). The first time I observed this behavior, I thought I had a new bee disease, until I realized that I was witnessing VSH behavior in action! You can observe it by looking for white-, or purple-eyed pupal faces being uncapped, and the healthy white pupae within often being chewed away. Dee Lusby has been reporting on this behavior for years: (Broken Link!) www.beesource.com/pov/lusby/lusbyjun.1997.htm. This behavior usually doesn’t kill the foundress mite, but sure keeps her from reproducing! You may ask, “But doesn’t chewing out brood weaken the colony?” Answer: it could, but the cost of removing brood is insignificant as long as mite levels remain low. For more information on VSH, see: http://www.glenn-apiaries.com

A dark VSH breeder queen (yellow queens are also available) from Glenn Apiaries. Photo Glenn Apiaries.

Figure 1. Illustration of varroa sensitive hygiene (VSH). Workers uncap cells to thwart reproduction by the mite. The beekeeper will observe exposed white- and purple-eyed pupal faces. The pupa may or may not be chewed out in the process.


Strong VSH in action. Note how the bees will remove the cappings over purple-eyed stage pupae, but may not remove the pupae. One bee in the upper right quadrant is chewing out a pupa.
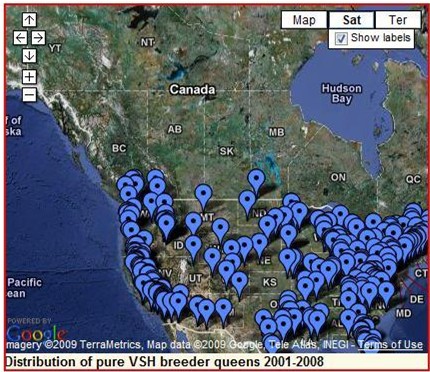
The good news. Tom Glenn has made this map of locations to which he has sent VSH breeders. We are slowly incorporating these genes into the U.S. bee population!
As far as my personal experience, the initial purebred VSH queen mothers I purchased were so inbred, that it was hard to keep their colonies alive, and the workers wanted to supersede them. The F1 crosses were better, but not fully mite resistant, nor great bees. However, two years ago, I ordered an instrumentally inseminated Minnesota Hygienic x VSH queen from Tom Glenn; she is the best queen I’ve seen since before varroa! I call her my “Dream Queen.” This particular cross of the individual parents was a magical combination. She’s been going two and a half years with absolutely no treatment, in a survival yard with colonies collapsing around her, and has negligible mite levels. Her open-mated daughters were the healthiest, strongest, best honey producers I’ve seen, and outperformed all other stocks stuck late in the mud at the end of last year’s rain-drenched almond pollination season. The Dream Queen colonies came out fat and happy, while everyone else starved. Needless to say, I’ve grafted a lot of daughters off her! Bee stock like this is the future of our industry; if all my colonies performed like hers, it would be like beekeeping in “The Good Old Days” before varroa again!

My “Dream Queen”–perhaps the best queen that I’ve ever had. She was a Minnesota hygienic x VSH cross from Tom Glenn, and lasted nearly three full years (finally was superseded after I wrote this article). Mite levels remained negligible without treatment, yet the colony (and her daughter colonies) were by far the most productive in my entire operation. The experience of having a colony like this shows me that breeding is the key to future successful beekeeping!
I can hear the skeptic saying “Yeah, but that’s how it was when Apistan worked.” True, but you’re missing the beauty of biological control: a chemical Silver Bullet can’t evolve with the mite; mite-resistant bees should be able to continue to handle varroa by evolving in lockstep with it. VSH, for example, is a great trait with which bees can fight the mite, but it’s not the only one. The bees have some more tricks up their sleeves…
Other potential varroa resistance/tolerance mechanisms
Here are some more observed or potential varroa-fighting tools likely to be found in the bees’ genetic toolbox (in rough order of likelihood):
1. Other hygiene-related behaviors (Dr. Marla Spivak is studying these). Apis cerana is so effective at VSH that Varroa jacobsoni generally won’t even try to reproduce on worker brood, even if transferred to worker brood of the European honey bee. (Boot, et al. 2004).
2. Grooming behaviors—there may be several genes involved:
a. Better autogrooming behavior to dislodge mites—especially useful with screened bottoms,
b. Allogrooming (nestmates grooming each other) behavior, and
c. Signaling or communicating the need to be groomed to nestmates,
d. Crushing of mites in the mandibles—Apis cerana and the Africans do this.
3. Resistance to various viruses and better immune system. It’s very important for the bees to keep the mite population in check, but it’s usually not the mites that cause colony collapse—it’s viral infection (notably deformed wing virus) vectored or initiated by mite feeding. Miaoqing, et al. (2005) state: “parasitization by varroa suppresses the immunity of honey bees, leading to activation of persistent, latent viral infection.” Surprisingly, the Honey Bee Genome Project (Evans 2006) found that compared to flies, honey bees possess only a third as many immunity genes. We should screen for bees naturally resistant to various viruses and other diseases.
4. Post-capping duration: Varroa is not well adapted to EHB worker brood—only 38% of the second female eggs are able to develop to maturity, and only 13% of the third. Theoretically, if the developmental period of the worker were shortened by one day, no third daughters would mature; by two days, very few second daughters (Figure 2). This has not yet been demonstrated by experiment, although it is often cited as a possible mechanism used by the African honey bee. Dee Lusby demonstrated that by selective breeding, one could knock up to 4 days off EHB queen development time (DeGrandi-Hoffman, et al. 1989), implying that worker development time could also be curtailed. Steve Taber suggested a selection method in the November 2006 ABJ.
5. Minimal drone production, since most mite reproductive success takes place in drone brood.
6. Biochemical—mites incorporate some bee proteins unaltered into their eggs without digestion (Tewarson and Engles 1982). Bees might evolve proteins that are deleterious to the mite; or the bee could modify essential proteins that varroa require for reproduction or metabolism.
7. Alteration of bee larval or pupal pheromones/volatiles that initiate mite reproduction or feeding (Denis Anderson in Australia, Yves Leconte, and Peter Teal are all working on this). It is necessary for the mite to “read” the pheromonal signal of a pupa to tell it to ovulate. Our problem mite—the Korean haplotype of Varroa destructor—mutated to be able to ovulate in response to worker, as opposed to drone, pheromones. We could breed bees that don’t give this signal, and would thus suppress mite reproduction in worker brood.
8. Dr. Spivak suggests that our breeding for bees that use less propolis may have backfired against us, since propolis may be the bees’ first line of defense against diseases, and possibly mites.
9. Thickened drone brood cappings to trap mites with multiple-infested drones, as in A. cerana.
10. The aforementioned mechanisms are only those that come to my mind. There are likely more—my guess is that we’ve only scratched the surface. A bibliography of research on varroa resistance can be found at: (Broken Link) https://scientificbeekeeping.com/wp-admin/www.glenn-apiaries.com/bibliog.html
11. We must also continue to breed for the usual traits of vigorous broodrearing, honey production, and gentleness, and resistance to tracheal mite and other diseases.
12. Add to the above list that we may wish to screen for virus-free queen lines (as with strawberries and horses). Viruses may lie latent in queens, and be passed on to eggs (Chen, et al. 2006). Furthermore, the manner in which queens are raised may be important. The new science of epigenetics (Watters 2006) has found that nutrition and stress events affect not only the individual, but also the behavior and disease resistance of its offspring, sometimes for several generations. In other words, if a queen is reared under stress or perforated by a mite, she may pass effects on to her offspring.
Wow, breeding for the best mite-tolerant queen is quite an order! Obviously, no breeder is going to investigate, let alone breed, for all these traits. It’s hard enough to breed for just a few! Plus, who’s to say what the best combination is? Mother Nature uses trial and error and many generations to determine which combinations are most efficient. Just as there are many recipes for the “perfect” chocolate cake, there may be innumerable recipes for the “perfect” bee. But we can certainly support researchers who dedicate themselves to the quest, and breeders such as Sue Cobey and Tom Glenn who maintain genetic banks from which breeders can draw stock to experiment with.
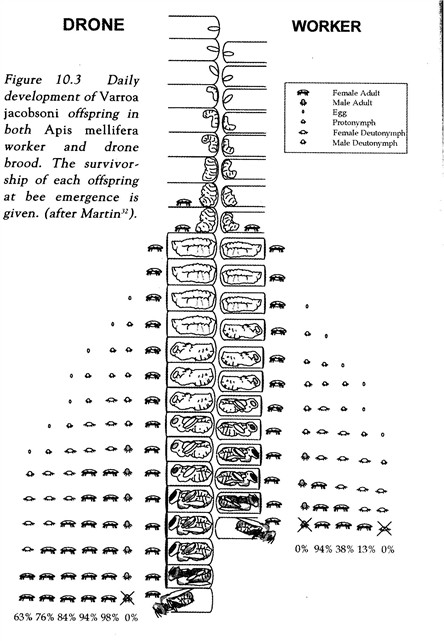
Figure 2. Varroa development in drone and worker brood. By breeding for bees with shorter post-capping development time, we may be able to restrain the rate of mite reproduction.
The Russian Bees
Another option is the “Russian Bees.” Dr. Tom Rinderer of the ARS Baton Rouge lab traveled to the Primorsky region of Russia to where European honey bees have been longest in contact with the varroa mite. He made trips to the region from 1994-2002 to collect promising stock (Broken Link) (www.ars.usda.gov/Services/docs.htm?docid=6444), and has now given the resultant lines over to collaborators in the Russian Bee Breeders Association (www.russianbreeder.org).
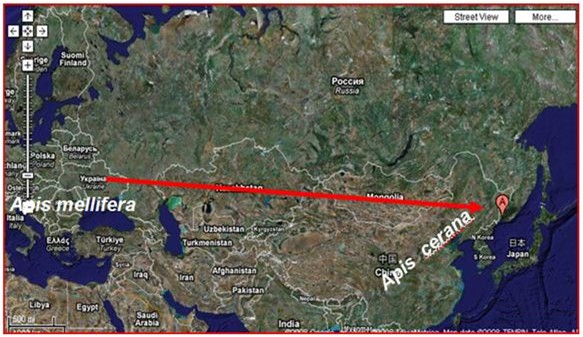
European bees from the Ukraine were moved to the Primorsky region about 150 years ago, where they came in contact with varroa.

I was able to spend some time with Dr. Rinderer and Russian breeder Carl Webb, in Carl’s yard in Georgia. The bees were well behaved, productive, and free of diseases. None of the breeders apply any treatments to their colonies.

The Russian bees aren’t “One Trick Ponies.” Here’s a list that I made of some of the mite resistance tactics that they use. When they shut down broodrearing, they actively scour mites out of the hive. This response to pollen flows may make Russians more suited for stationary, as opposed to migratory, beekeeping.
The Russian bees winter very well, in small clusters, and build up explosively in response to pollen flows in spring (or fall). They are noted for how quickly they will fill the supers, despite having smaller populations than Italian bees.
I’ve spoken to a number of beekeepers who swear by purebred Russian stock. Please note that Russian hybrids may not perform nearly as well as the purebreds.
Genetic diversity of bee stocks
We may have a problem: loss of genetic variation in our bees. Genetic variation is the list of “ingredients” available in a species from which to concoct evolutionary adaptation, or to use in selective breeding by humankind. There may be a paucity of genetic stock due to several factors:
1. The U.S. cut off imports of honeybees in 1922 (due to tracheal mite problems in Europe), leaving us with only 7 subspecies of Apis mellifera (of the 26 known in the world) to breed from. Of these seven, commercial stocks generally are based upon only three!
2. Then we lost about 70% our bees when tracheal mite finally arrived in 1984,
3. and then lost an even higher percentage when varroa arrived in 1987. As if that weren’t enough,
4. Debby Delany (Washington State Univ) surveyed queen producers and found that in the mid ‘90’s only 603 breeder queens produced one third of all production queens used in the country!
This lack of diversity is called a “genetic bottleneck,” and means that our bees didn’t have many genetic tools to work with to fight varroa. Progressive breeders, such as Sue Cobey and Steve Sheppard, and Tom Rinderer (Broken Link!) (Russian, www.ars.usda.gov/Services/docs.htm?docid=6444), are bringing in other genetic lines. Others, such as Joe Waggle are selecting from feral colonies
(Broken Link!) http://pets.groups.yahoo.com/group/FeralBeeProject/, in the hope that they possess traits not found in commercial stocks.
Update: further research calls into question whether the U.S. is indeed lacking in bee genetic diversity. I will cover this in a future article.
Diversity within the colony
An important aspect of bee colony behavior and disease resistance is that a colony of bees is composed of subfamilies of “supersisters,” all having the same mother (the queen), but each subfamily having a different drone father (due to the queen’s multiple mating). In a colony, there can exist one group of supersisters really good at honey production, and another group really good at VSH, and another group that excels at comb building. Together, they work as a functional unit superior to any single subfamily of supersisters alone. Tarpy and Seeley (2006) found that colonies headed by multiply-mated queens were more resistant to AFB than those whose queens were inseminated by only one drone. Everything about honeybee mating behavior is geared toward multiple matings to ensure a large number of “patrilines” present in each colony. We may wish to emulate this natural scheme in our breeding programs.
I got lucky with my Dream Queen, in that I apparently got a perfect mix of supersisters who together, “had it all;” but it made me realize that our future may lie in returning to programs like the Starline bee. Witherell (1976) describes the maintenance of the famous Starline and Midnight queens developed by Dadant. They were each double (line) “hybrids” or “crosses” of four inbred lines specifically chosen for their “combining ability” to produce exceptional and predictable production queens. Collaborating producers bought Line AxB (Line A queen inseminated by Line B drones) on even years, and Line CxD on odd years. That way, the previous years’ queens would produce drones with genes derived from, say, A and B lines to mate with virgin queens from this year’s CxD cross. The next year, the inverse would hold true.
A queen producer (as opposed to a queen breeder) could do something similar today. If he/she produced queens from Minnesota Hygienic x VSH stock this year, their colonies could supply drones next season for a breeder queen of say, Russian x New World Carniolan parentage (I have no idea if any of these particular hybrids would have “combining ability”—I’m just proposing an example). If that double hybrid were successful, the following year the queen producer could purchase another MH x VSH to repeat the cross.
Breeding your own stock
You can shift your operation to varroa tolerance by purchasing selected queenstock, or you can start from your own locally-adapted stock (paying special attention to surviving feral colonies). Erickson, et al. tell you how to do it: “Producing Varroa-tolerant Honey Bees from Locally Adapted Stock: A Recipe” (Broken Link!) www.beesource.com/pov/usda/varroatolerantbee.htm. In England, Roger Dewurst selects for bees that bite the beekeeper by tugging at his skin! He finds that the amount of grooming and damage to mites increases in the subsequent generations (see photos at : (Broken link!) www.kilty.demon.co.uk/beekeeping/improvement.htm).

Many beekeepers are intimidated by the prospect of queenrearing. However, once you try it, you will be amazed at how easy it is! Here I’m pulling out a frame of queen cells from a queenright cell finisher colony.
Some breeders, such as Steve Taber and John Kefuss, can be downright brutal—using the so-called “Bond” method (Live and Let Die) (Kefuss 2004), whereby your best colonies (he suggests starting with freeze-kill tested hygienic bees) are placed into a survival yard, without treatment, to see who survives. As the losers collapse, they infest the survivors with additional mites. You could call it the “acid test,” but any surviving colonies would be worth breeding from. I personally set up a Bond survival yard, but seeing almond pollination prices pushing $150 last year, I chickened out in late summer, and gave some of the surviving colonies a “soft” mite treatment (but removed them from the breeding pool). This made me realize that we shouldn’t count on western queen producers to perform many survivability trials, since they also depend upon pollination income for their livelihoods! They may have to look for breeding stock from eastern beekeepers who can allow yards to crash, and then make up their winter losses each spring from the survivors. A reasonable compromise is practiced by a number of beekeepers: they just help the bees once or twice a year, say, with a shot of oxalic acid on the broodless winter cluster, so they get a fresh start each season, then breed from their best surviving colonies each spring.
What I’m personally doing now is testing all my colonies at the same time each August with sticky boards. Those that have extremely low mite levels (after checking to make sure the colony is populous and productive) are marked as potential breeders, not treated, and watched. If they make it ‘til fall with low levels, they are again checked for condition when I prepare for almonds in late January. The best are put aside for breeders, and the rest get extra drone combs to fill with drones in the almonds, so they’ll be good “drone mothers” when they return home. All other colonies that needed miticide help, have their drone brood removed by trap frames in February and March (so they don’t contribute to matings), and are nuked up (killing the queens) to repopulate my operation. I add a few mite-resistant inseminated queens each spring from Tom Glenn and elsewhere, to maintain genetic diversity, by serving (if they prove themselves) as drone or queen mothers next season.

Breed only from healthy, productive colonies that maintain low mite levels.
Update 2009: “Survivor” stock may handle varroa, but may not be productive enough to be worth keeping. Breeding solely from survivors may lock in certain key alleles for mite resistance, but this avenue may be best left to professional breeders and Mother Nature. In your own operation, I suggest that you only breed from productive colonies, and add resistance alleles via purchased drone mother queens.
If you raise your own queens, it will likely take at least three years to shift the genetics of your operation (since you must also shift the genetics of the feral colonies that send out drones) to the point that you see a major difference.

The genes of the virgin queens in these cells are important, but half the genetics of the resulting colonies will come from the drones that the queen mates with.
Your turn
Spring is just around the corner, and with it comes time to requeen colonies. The first step to an IPM program against varroa is to start with mite-resistant queens (Broken Link) (www.glenn-apiaries.com/queenproducers.html). If you’re adventuresome, and can afford having most of your colonies die, you could start from scratch. However, it would be much easier and quicker to begin with existing mite-tolerant commercial stocks. Demanding mite-tolerant queens from a producer is like buying tomato plants resistant to verticillium wilt. There is no sense in purchasing plants that are likely to die, even with babying and chemical treatments, when there are varieties available that can thrive on their own! It’s the same with bees. Ask your queen supplier what treatments he or she uses in their own operation to keep their bees alive. Or better yet, ask how long his/her breeders have gone without any treatments at all for varroa? As purchasers of queens, you have the opportunity to change the market. By demanding mite tolerance in the queens you buy, you can shift the genetic structure of the bee gene pool in North America!
WARNING: it is unlikely that simply requeening with mite-resistant stock will end your mite problems at first! There are other factors involved. Keep in mind that your resistant bees will still have to deal with the onslaught of mites from collapsing colonies in your vicinity until other local beekeeping operations and the feral population shift toward mite resistant genetics. At some point we will reach a “tip point” where the majority of all colonies are mite resistant, and we won’t be picking up the fallout from the chemical-coddled collapsing colonies of our neighbors.
There is also the question as to whether the queens you purchase are well adapted to your area and operation—performance and seasonal behavior of various resistant stocks vary greatly. For example, one of the ways that Russian stocks deal with mites is to overwinter with a small cluster. This may be a problem if you want huge colonies in February for almond pollination. However, there is no reason to think that mite resistant bees can’t perform well in migratory commercial operations, as long as one breeds from the best performers each season.
Don’t expect a miracle! But remember, we don’t necessarily need bees that are 100% resistant to varroa—even bees that can delay the interval between mite treatments are valuable. Varroa opened up a Pandora’s Box of problems for our bees—stress from being fed upon, weaker wintering, virus and disease issues. Having bees that can keep mite levels lower will help them to overcome all those problems. For some period of time, though, you will likely need to help your bees with the battle. You will want to monitor mite levels, and have backup plans for mite control in place. In coming installments of this series, I will detail your options.
Those of us who were keeping bees prior to varroa remember how much easier it was back then. It is my fervent belief that we can return to that kind of beekeeping once we keep bees that handle varroa on their own!
Update: I will be greatly expanding this section on breeding soon!
References
Boot, et al. 2004 Natural selection of Varroa jacobsoni explains the different reproductive strategies in colonies of Apis cerana and Apis mellifera. Exp. & Applied Acarology 23(2): 133-144.
Chen, Y. P., Pettis, J. S., Collins,A. and Feldlaufer, M.S. 2006 Prevalence and Transmission of Honeybee Viruses. Appl Environ Microbiol. 72(1): 606–611.
DeGrandiI-Hoffman,G., Lusby, D., and Erickson, E. 1989 Managing Colony Genetics by Grafting and Selecting for Queens With Shorter Development Times. ABJ 129 (11): 717-719.
Evans, et al. 2006 Immune pathways and defence mechanisms in honey bees Apis mellifera. Insect Molecular Biology 15(5): 654-656.
Kefuss, J., Taber II S., Vanpoucke J. and Rey F. 2004. Breeding for varroa resistance: How we do it. ABJ July 2004
Miaoqing Shen, Xiaolong Yang, Cox-Foster, Diana, Liwang Cui. 2005 The role of varroa mites in infections of kashmir bee virus (KBV) and deformed wing virus (DWV) in honey bees. Virology 342(1): 41-149.
Spivak and Reuter 2001 Varroa destructor infestation in Untreated Honeybee (Hymenoptera: Apidae) Colonies Selected for Hygienic Behavior. J. Econ. Entomol. 94(2): 326-331.
Sheppard, S. 2006 Selection and possibilities within honey bees – be careful what you are selecting for. www.beeculture.com/storycms/index.cfm?cat=Story&recordID=480
Tarpy, D. R. and T. D. Seeley. (2006). Lower disease infections in honeybee (Apis mellifera) colonies headed by polyandrous vs monandrous queens. Naturwissenschaften, 93: 195-199.
Tewarson, T. & Engles, W. 1982. Undigested uptake of non-host proteins by Varroa jacobsoni. J. Apic. Res. 21: 222-225.
Watters, Ethan. 2006. DNA Is Not Destiny. Discover Nov. 2006, pp. 32-37, 75.
Witherell, Peter 1976 A Story of Success—The Starline and Midnite Bee Breeding Programs, ABJ 116(2): 63-64, 82.



