Probiotics and Colony Productivity
January 29, 2017
Probiotics and Colony Productivity
Randy Oliver
ScientificBeekeeping.com
First Published in ABJ in February 2016
My apologies for skipping a month in this colony microbiota series—too much bee work and too many conferences to attend. But before I return to beebread, I’d like to share the results of a related study of my own.
Probiotics
As we learn more and more about the symbiotic microbial communities that reside in or on the bodies of many organisms (including humans and bees) the more we find how little we know about their contributions to our digestion, health, immunocompetence, and even mood. Scientists have shown that in some cases, that by consuming certain symbiotic bacteria, one may be able to “retune” the gut to a healthier state, or in the case of livestock, to improve health and productivity [1]. Such beneficial microorganisms, which when administered to a person or animal in order to confer a health benefit are called “probiotics.”
Another term that you may hear is “prebiotics.” These are nondigestible food ingredients that promote the growth of the beneficial microorganisms in an animal’s gut.
Application To Beekeeping
As I’ve detailed previously, the honey bee is host to a “core” bacterial community that resides its gut, as well as other bacteria that colonize its hypopharyngeal glands and beebread. These bacteria appear to be involved in digestion [2] and disease resistance [3], and may produce critical nutrients not found in nectar or pollen [4]. Not surprisingly, researchers have proposed that colony health may be improved by probiotic inoculation [5].
Acroynym-loving scientists have created new terms to address colony “dysbiosis” (microbial imbalance)–Microbial Resource Management (MRM) and Symbiont Resource Management (SRM–defined as “the application of microbial symbionts to manage insect-related problems” [6]). Entrepreneurs of course have seized the opportunity to sell something new to beekeepers, and are currently marketing probiotic products. The question, of course, is whether their claims can be substantiated [7].
Potential Modes Of Action
There are various ways in which probiotic/prebiotic products could potentially improve bee health. What is important to understand is that the microbial community in the gut is continually changing. Each variation in food (e.g., almond pollen vs. dandelion pollen) may introduce or favor one strain of microorganism over another. Add to that the effects of antibiotics, pesticides, and natural antibacterial plant metabolites–any of which may cause dysbiosis (think of your gut after a course of antibiotics).
A different strain of bacteria coming in with nectar or pollen may gain a foothold and outcompete some established members of the gut biota (think of Montezuma’s Revenge). And the short generation time of gut bacteria (15 minutes to hours), plus their ability to swap plasmids of genetic information between species, allows them to continually evolve in their competition for food and habitat in the gut. Metagenomic characterization of the microbial community structure of the bee gut indicates that, despite there being a few core species, it can be rather variable.
That said, the feeding of a prebiotic could potentially favor certain beneficial bacteria. This may especially applicable to the formulation of pollen subs.
Probiotics, on the other hand, consist of living bacteria. One could make the case that they could help to reestablish the core gut community after treatment with antibiotics. However, it appears that the gut biota of commercial bee stocks are already resistant to antibiotics [8]. And a field study did not find any detrimental effect from starting off packages with a dose of multiple antibiotics [9].
In any case, it is certainly plausible that feeding a probiotic containing the right strains of bacteria could help to bring the hive microbial community into tune.
The Importance Of The Right Bacteria
Feeding live bacteria willy nilly to bees may not be the best idea. Even nonpathogenic bacteria may cause a metabolically costly upregulation of their immune response [10]. Bacteria that exhibit a salubrious effect in one species (say humans or livestock) may not be of any benefit to the honey bee. Indeed, introducing the wrong probiotic bacteria can worsen colony health. Most likely, any probiotic for bees should consist of one or more specific bacterial strains originating from the guts of healthy bees.
Practical application: dumping pet probiotics or yogurt into your hives is likely a waste of money.
Have Probiotics Been Shown To Be Of Benefit?
Probiotic treatments are offered by the bee supply companies, and a number of beekeepers have experimented with various animal probiotics. Unfortunately, some of the claims for such products may be based more upon wishful thinking than actual scientific testing. Indeed, there is a paucity of studies that have actually demonstrated any beneficial effects from feeding probiotics to honey bees.
Since fumagillin is not allowed to be used for nosema management in Poland, researchers there have run a number of trials of probiotics and prebiotics, in general, finding generic veterinary formulations to be of little or no observable benefit [11].
When he heard of California beekeepers feeding a pet store probiotic to their bees, Dr. Eric Mussen included it in a recent trial [12]. The result: “This study did not find a beneficial role for the feeding of probiotics to honey bee colonies.”
More Harm Than Good?
Feeding the wrong probiotic bacteria to bees can cause more harm than good. Andrearczyk [13] observed greater mortality of caged bees when they were fed an animal probiotic mixture. Of serious concern to kitchen cowboys is that the wrong probiotic or prebiotic can make bees more susceptible to nosema. Ptaszyńska [14] recently found:
In conclusion, the supplementation of honeybees’ diet with improperly selected probiotics or both probiotics and prebiotics does not prevent nosemosis development, can de-regulate insect immune systems, and may significantly increase bee mortality…mortality rates increased among honeybees fed with commercial probiotic containing L. rhamnosus. Therefore, preparations containing bacteria identified as probiotics for mammals should not be considered as probiotics for honeybees.
Practical application: don’t just go dumping any bacterial probiotic into your hive.
On the other hand, using specific bacterial strains cultured directly from the guts of healthy bees may indeed show potential benefit [15].
A Study Of My Own
Back in 2012, after finding a distinct difference in Nosema ceranae levels between my productive vs. unproductive colonies [16], it occurred to me that a similar sort of screening might be an easy way to see whether productive colonies harbored a different bacterial gut community than did nonproductive hives. No matter which way the results went, I’d learn something:
- If there was a consistent difference, then I would have identified which bacteria were associated with greater productivity.
- On the other hand, if there was no clear correlation between productivity and microbial structure, I’d be less enthusiastic about the premise that treating colonies with a probiotic would improve productivity.
Scientific note: I prefer to run simple “quick and dirty” field or lab studies, and to publish my findings in ABJ rather than scientific journals, since I can get useful information out to the beekeeping community in real time (sometimes publishing within a month of completing a trial). This is opposed to the tedious process of working with institutions and going through peer review (which I strongly support [17]), but which typically takes 2-3 years to get the results published—as it did in the case of this study, which my collaborators and I recently published in the journal PeerJ [18]. Following is a condensed version of the paper written specifically for beekeepers.
Introduction
As with other livestock producers, there is interest in the beekeeping industry as to whether it would be of benefit to feed probiotic bacteria to honey bee colonies. Any potential benefit would presumably be the result of the colonization of the bee guts by the introduced probiotic. The simplest metric of benefit would be increased honey production, since it directly reflects overall colony health.
By extension then, and in order to select specific bacteria for use as probiotics, there should be evidence that a difference exists between the gut microbiota of productive and nonproductive colonies in the field.
Materials And Methods
During a rough summer in the drought-ravaged California Sierra Foothills, when we went to pull honey, we found (not unusually) stark colony-to-colony differences in production, even accounting for overall colony strength (and despite the fact that we establish each yard with fresh nucs each spring, generally all from only one or two queen mothers per yard).
Buoyed by my recent success with the nosema screening, I contacted Dr. Irene Newton, who had recently published research using metagenomics to map the bee gut microbial communities, and proposed a study. We agreed on a sampling procedure, and I used beekeeper donations to ScientificBeekeeping to pay for the analysis, which was undertaken by her grad student Melissa Horton.
In August, after the end of the honey flow, hefted and inspected every hive in each of 7 yards of approximately 24 hives each in order to determine for each location the two hives that had put on the most honey (productive), and the two that had gained the least (nonproductive). We excluded from the nonproductive group any colonies that were not strong with bees (to eliminate the effect of colony strength), or that showed any signs of disease or other impairment (Fig. 1).
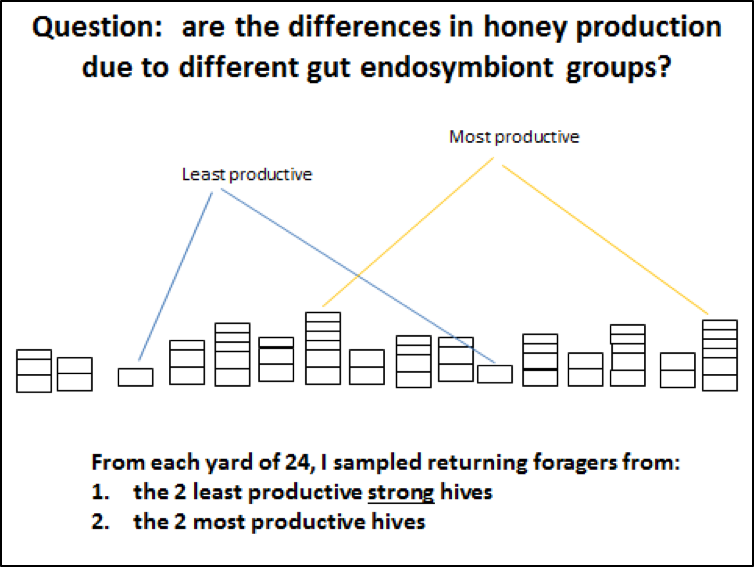 In order to standardize the bees sampled, we collected returning pollen foragers, based upon Dr. Newton’s previous findings that their gut microbiota was typically representative for the colony. We intercepted incoming foragers with pollen loads, humanely dispatched them, and then used sterile forceps to gently tease out their entire hind and midguts (Fig. 2). We collected a minimum of 5 samples from each hive, preserving them immediately into RNAlater®, chilling them on ice, and then freezing until analysis, which was performed at Indiana State University by Melissa Horton.
In order to standardize the bees sampled, we collected returning pollen foragers, based upon Dr. Newton’s previous findings that their gut microbiota was typically representative for the colony. We intercepted incoming foragers with pollen loads, humanely dispatched them, and then used sterile forceps to gently tease out their entire hind and midguts (Fig. 2). We collected a minimum of 5 samples from each hive, preserving them immediately into RNAlater®, chilling them on ice, and then freezing until analysis, which was performed at Indiana State University by Melissa Horton.
 Melissa extracted the bacterial DNA from the samples and Illumina sequenced it to identify the bacteria present [19].
Melissa extracted the bacterial DNA from the samples and Illumina sequenced it to identify the bacteria present [19].
Results
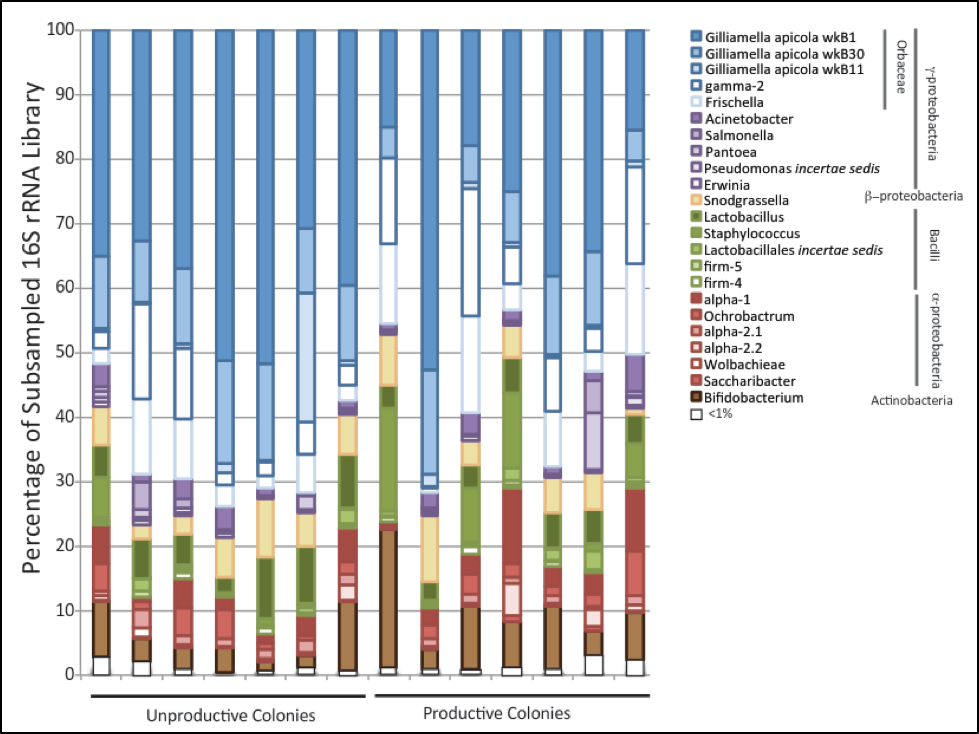
In this case, a picture is worth a thousand words (Fig. 3).
Simple visual comparison of the distribution of the colored bars between the nonproductive colonies (to the left) vs. that of the productive colonies (right), indicates that there was no obvious bacterial community structure associated with higher productivity. So Melissa performed state of the art analysis of the data in various ways.
Scientific note: sometimes in a complex data set as above it is difficult to distinguish patterns that occur in multiple dimensions. So we perform discriminate function analysis, which in this case could tell us whether colony productivity was associated with specific community structures.
Discriminant function analysis could readily discriminate productive and unproductive colonies. Surprisingly, two bacteria [20] which occurred at extremely low frequency were good predictors. Although they are unlikely to have contributed functionally, they could perhaps be indicators of colony microbiome diversity or evenness. In general, productive colonies exhibited greater bacterial diversity, richness, and evenness.
Initial in silico sampling appeared to strongly differentiate between the groups, but this was not supported by biological sampling. In order to test for what may have been a competitive exclusion of other bacteria by Gilliamella apicola, Melissa cultured them, but found no evidence of a negative interaction.
Discussion
Although we found that productive colonies trended towards increased diversity and prevalence of Lactobacillus species (such as Firm-4), and were less likely to be dominated by Gilliamella species, these trends were not consistent enough to suggest that they were the main reason for increased productivity. Perhaps with a greater number of samples, we could have found a statistically-significant trend, but there was clearly no black or white difference between the groups, and no single bacteria stood out as being particularly “beneficial.”
Since there was “no apparent correlation between honey bee forager gut microbiota and honey production” under natural conditions (in the Sierra foothills), our results do not support the hypothesis that colony productivity would be improved by the feeding of specific probiotic bacteria. However, the sampled colonies had not been exposed to antibiotics or ag chemicals (since almond pollination in February), and all sampled colonies would have been considered as being “healthy.” This leaves the door open for further research.
As far as correcting suspected colony gut “dysbiosis” by the feeding of a probiotic supplement, one should keep in mind the constant input of fresh bacteria into the “common stomach” of the colony via the foragers, as well as the high rate of food exchange between the nurses and all other bees in the hive [21]. Such exchange would serve to reinforce microbiome similarity throughout the hive, and would require any artificially-introduced probiotic strain to compete against at least some of the resident microbiota.
It seems unlikely that a repurposed human, pet, or livestock probiotic would be able to establish itself to the benefit of the colony, and as has been previously noted, may indeed create its own dysbiosis. On the other hand, if bee-specific strains can be commercially cultured, it’s possible that someone may develop a useful probiotic formula. But I caution beekeepers to ask to see strong data to back up any marketing claims.
Acknowledgements
I wish to express my appreciation to Dr. Irene Newton and Melissa Horton for their hard work and patience during our multiple drafts of the published paper. Thanks to the donors to ScientificBeekeeping for their funding of this study. And to beekeeper Brion Dunbar for his assistance in collecting samples. And of course to Peter Borst for his unflagging assistance in literature search.
Footnotes and Citations
[1] For example, Bäckhed, F, et al (2004) The gut microbiota as an environmental factor that regulates fat storage. http://www.pnas.org/content/101/44/15718.full.pdf+html
[2] Lee, FJ, et al (2014) Saccharide breakdown and fermentation by the honey bee gut microbiome. Environ Microbiol 17(3):796-815.
[3] Alippi, A.M. and Reynaldi, F.J. (2006) Inhibition of the growth of Paenibacillus larvae, the causal agent of American foulbrood of honeybees, by selected strains of aerobic spore-forming bacteria isolated from apiarian sources. J. Invert. Pathol. 91, 141–146
Sabate´, D.C et al. (2009) Inhibition of Paenibacillus larvae and Ascosphaera apis by Bacillus subtilis isolated from honeybee gut and honey samples. Res. Microbiol. 160, 193–199
[4] Dillon, RJ & VM Dillon (2004)The gut bacteria of insects: Nonpathogenic Interactions. Annu. Rev. Entomol. 49:71–92.
[5] Hamdi, C, et al (2011) Gut microbiome dysbiosis and honeybee health. J. Appl. Entomol. 135: 524–533.
[6] Crotti, E, et al (2013) Microbial symbionts of honeybees: a promising tool to improve honeybee health. New Biotechnology 30(6): 716–722.
[7] Rijkers, GT, et al (2011) Health benefits and health claims of probiotics: bridging science and marketing. British Journal of Nutrition http://library.wur.nl/WebQuery/edepot/177309
[8] Tian B, et al (2012) Long-term exposure to antibiotics has caused accumulation of resistance determinants in the gut microbiota of honeybees. mBio 3(6):e00377-12. doi:10.1128/mBio.00377-12
[9] Johnson, BR, et al (2014) Effects of high fructose corn syrup and probiotics on growth rates of newly founded honey bee colonies. Journal of Apicultural Research 53(1): 165-170
[10] Evans JD & DL Lopez (2004) Bacterial probiotics induce an immune response in the honey bee (Hymenoptera: Apidae). J Econ Entomol. 97(3):752-6.
[11] Most of the below are open access:
Kaznowski, A, et al (2005) The effect of probiotic supplementation on the content of intestinal microflora and chemical composition of worker honey bees (Apis mellifera). J Apic Res 44(1): 10-14. Apparent slight beneficial effect from a single initial feeding.
Kazimierczak-Baryczko, M & B Syzmas (2006) Improvement of the composition of pollen substitute for honey bee (Apis mellifera L.), through implementation of probiotic preparations. Journal of Apicultural Science 50(1): 15-23. This study showed suggestive evidence of benefit.
Pătruică, S, et al (2013) The effect of prebiotic and probiotic products used in feed to stimulate the bee colony (Apis mellifera) on intestines of working bees. Journal of Food, Agriculture & Environment Vol.11 (3&4): 2 4 6 1 – 2 4 6 4
Although colonies fed with pre- and probiotically modified feeds [lactic or acetic acid; human probiotic mixture] showed enhanced development during the first part of the season, in the autumn the number of combs covered with bees and the reserves of food for winter were comparable with those found in the control group.
[12] Johnson, BR, et al (2014) Op cit.
[13] Andrearczyk S, et al (2014) Influence of a probiotic on mortality, sugar syrup ingestion and infection of honeybees with Nosema spp. under laboratory assessment. Med Weter 70:762–76. (Broken Link!) http://medycynawet.edu.pl/images/stories/pdf/pdf2014/122014/201412762765.pdf
[14] Ptaszyńska, AA, et al (2015) Are commercial probiotics and prebiotics effective in the treatment and prevention of honeybee nosemosis C? Parasitol Res DOI 10.1007/s00436-015-4761-z Open access.
[15] Audisio, M & M Benítez-Ahrendts (2011) Lactobacillus johnsonii CRL1647, isolated from Apis mellifera L. bee-gut, exhibited a beneficial effect on honeybee colonies. Beneficial Microbes 2(1): 29-34.
[16] https://scientificbeekeeping.com/nosema-ceranae-and-honey-production-in-healthy-colonies/
[17] But don’t get me started on the deplorable state to which the peer review process has depreciated in recent years, as scientists desperately publish anything to which they can give a “sexy” title to justify their funding or position.
[18] Horton, MA, Oliver, R and IL Newton (2015) No apparent correlation between honey bee forager gut microbiota and honey production. PeerJ3:e1329; DOI10.7717/peerj.1329
[19] See the original paper (ibid) for details.
[20] Bacilli incertae sedis and Comamonadaceae incertae sedis (insertae sedis means that their taxonomic classification is not yet clear).
[21] As well explained by: Kapheim KM, et al (2015) Caste-specific differences in hindgut microbial communities of honey bees (Apis mellifera). PLoS ONE 10(4):e0123911 DOI 10.1371/journal.pone.0123911.