Sick Bees – Part 9: Pathogens and Plagues
June 3, 2011
Contents
Plagues
Emerging Infectious Bee Diseases
Models for Emerging Pathogens in Wildlife
Introduced Pathogens
The Next One
Viruses are Everywhere!
Alternative Hosts
How Do the Bees Keep Up?
The Four Horsemen
Bees as (Semi-) Domestic Animals
Coming to Terms with Colony Collapse
References
Sick Bees—Part 9 & 10
Pathogens and Plagues
Randy Oliver
ScientificBeekeeping.com
First published in ABJ May and June 2011
Our latest “bee plague,” Colony Collapse Disorder, may finally be losing steam—there were just about enough hives this year for California almond pollination. But it is only a matter of time before beekeepers get hit by the next plague. In this article I’d like to look at old, emerging, and future plagues of infectious bee diseases.
Plagues
The human population, as does the bee population, suffers from bouts of epidemic infectious diseases. Take the Bubonic Plague, caused by a bacterium vectored by fleas from rodents to humans. There have been three great waves of human Bubonic Plague in historical times, at times killing up to half the human population in struck areas. These waves came centuries apart, with the second wave continuing to strike every 10 years or so for nearly 300 years! The Plague would appear to go into hiding between bouts. Plague has hardly disappeared—there were epidemics in three continents in the 1980’s.
What strikes me are the many similarities between human plagues and bee plagues:
- They are generally fairly recently evolved or introduced: The Plague bacterium evolved from another species only a few thousand years ago, and was then introduced from China to the other continents. Varroa, and a number of our other bee pathogens are recently introduced or, in the case of viruses, new strains.
- They often evolve into a number of different strains: Similar to the bee viruses, each strain of Plague has slightly different characteristics (Achtman 2007), which may relate to its infectivity and virulence in its rodent and human hosts.
- They have an asymptomatic reservoir: Between outbreaks, the plague bacterium circulates within populations of certain species of rodents without causing excessive mortality. Such groups of infected animals serve as silent, long-term reservoirs of infection. Similarly, bee viruses normally exist as inapparent infections in bees and other insects.
- They can infect multiple hosts: The plague infects various species of rodents, and also cats, dogs, and some other mammals, and causes periodic epizootics, perhaps due to a slight mutation. Honey bee viruses also infect various other bees and wasps.
- They are transmitted by a vector: The Plague by fleas; bee viruses by both varroa and by drifting and robbing bees.
- They can cause periodic outbreaks: Note that epidemics may recur periodically for many years, and then disappear for a while.
- The outbreaks can be devastating: When an epidemic takes off, it can kill from half to all of the host population.
Emerging Infectious Bee Diseases
There is nothing unique about plagues in honey bees. They follow the model of epidemics in humans (and epizootics in wildlife). What we appear to be currently experiencing are the effects of several concurrent “emerging” (or “emergent”) infectious diseases in the bee population. Dobson (2001) defines “emerging (or re-emerging) infectious diseases [as those] whose geographical range, host range or prevalence have been increasing in recent years.” Nosema ceranae is an example of an emerging pathogen of the European honey bee (Higes 2010) since it jumped hosts from the Asian Bee to the European honey bee and is displacing its cousin, N. apis.
Other emergent bee pathogens are the mutable forms of the Kashmir Bee Virus family, and Deformed Wing Virus (Genersch 2010). Unlike N. ceranae, the viruses were already present (but largely unnoticed) in the bee population, but became emerging epidemic diseases when varroa mite changed their transmission and reproduction dynamics.
In the U.S., chalkbrood fungus, the tracheal mite, and the varroa mite have all been emerging pathogens within the last 45 years, as each arrived and then invaded the entire bee population, but all would now be considered to be established parasites, although only chalkbrood and the tracheal mite have reached a mature host/parasite relationship with the bees.
Models for Emerging Pathogens in Wildlife
We can learn what to expect with emerging pathogenic diseases in honey bees by studying epidemics in wildlife. I have lifted quite a bit of information for this article from two excellent sources—Dobson (2001) and Ostfeld (2008). Dobson studied 31 cases of emerging wildlife epidemics, and concluded that they could be placed into three categories:
- Recently invading parasites. “Because of high host susceptibility the introduction of novel pathogens is often followed by explosive spread through the host population in what is termed a ‘virgin ground epidemic.’ These pathogens most frequently originate from another host species (e.g., chalkbrood and the mites), but can also result from a mutation in a virus (as an example Dobson cites canine parvovirus, which spread throughout the world’s populations of dogs, coyotes, and wolves in only 15 years).
- “A second type of emerging pathogen is those native to a specific host and geographical region that are currently spreading within the host population as a result of new external factors.” The viruses would largely fall into this category, with varroa being the main new factor. But Dobson also raises an interesting point: “Alternatively, some environmental factors, like pollution, can stress hosts and reduce their ability to respond to existing parasite infections.” This may well be the case with regard to the problems that beekeepers have had when heavy miticide or pesticide contamination of combs allows opportunistic pathogens to decimate their colonies.
- “A final category of pathogens emerges as a result of a combination of the previously mentioned circumstances. Here we would include pathogens that have recently invaded an immunologically naïve host population that is in addition stressed or immunocompromised because of existing environmental conditions.” I introduced the importance of immunocompetence in my last article. Parasite epidemics typically explode when colonies are suffering from nutritional stress due to drought, rain, or placement on a monoculture. With the load of novel parasites that bees are carrying these days, they are even more susceptible to parasites when their immunocompetence is compromised by nutritional or chemical stresses.
Dobson then goes on to describe typical scenarios:
“Pathogens responsible for wildlife epidemics are frequently characterized by particular life-history traits that facilitate emergence. We found that the majority of these pathogens are microparasites that lack intermediate stages and have a direct life cycle [none of the bee pathogens have intermediate stages, as do tapeworms, etc.]…In addition, many emerging pathogens have catholic preferences regarding host suitability and can reproduce within a variety of related host species [as with bee viruses]. Emerging pathogens appear to have high transmission rates and are spread directly, either through contact between infected and uninfected hosts or indirectly through vectors [such as varroa]. Finally, some pathogens have the capacity to remain infective for long periods of time” [as is the case with the ubiquitous inapparent infections by the bee viruses].
“The majority of causative pathogens were of exotic or likely exotic origin…This suggests that it is more likely that an exotic pathogen will produce a severe wildlife epidemic than a pathogen locally co-evolved with its host [most of our serious bee parasites are of exotic origin]. Nevertheless, we did identify several examples of exclusively viral pathogens that apparently produce regular epidemics in wildlife populations” [as the bees viruses have historically done].
Interestingly, Dobson also found that humans were in some way involved in the majority of the outbreaks of epidemics in wildlife. Note that Dobson was studying wildlife, not domestic animals. Honey bees fall somewhere between wild and domestic, but humans are generally involved in bee epidemics, either by the transportation of bees and their parasites, or by creating additional stresses upon the bees: “Transported animals are often crowded and stressed and are therefore particularly susceptible to infection even if only a few individuals are infected.” This brings to mind the overpopulated winter holding yards for hives in California.
The take home message is that the episodic epidemics in honey bees are quite similar to those in other wildlife. In some cases, such as with Thai Sacbrood, the epidemics occur in regular waves. In other cases, as with nosema, Disappearing Disease, or Sacbrood, the episodes occur sporadically, and are generally associated with environmental factors, such as poor nutrition or chill events. There are also occasional outbreaks from what are apparently novel virulent mutations of some of the normally benign bee viruses, such as by Chronic Paralysis Virus or Kashmir Bee Virus—these generally burn out quickly, as do human epidemics of highly virulent viruses such as Ebola or Hantavirus.
Introduced Pathogens
As Dobson pointed out, most animal epizootics are due to exotic pathogens, meaning that they have either jumped hosts, or were introduced by humans carrying them across geographical barriers, such as mountain ranges or oceans.
So where would an emerging new pathogen of the European honey bee come from? The most obvious source is from our bees’ cousins, the other honey bee species, or from other bees or wasps. The Asian bee, Apis cerana (Fig. 1), was clearly the source of varroa, and likely also the source of some of the bee viruses, the tracheal mite, and Nosema ceranae. It’s possible that some Asian viruses required the presence of the mites in order to gain a foothold in the European honey bee.
Figure 1. The Asian Bee (Apis cerana) is the original host of varroa, and likely several other parasites that have jumped to the European honey bee, often with devastating consequences. Photo credit: Wikipedia.
So how did the parasites of Asian bees get across the Pacific Ocean to the U.S.? Well, two likely sources were the large scale importation of non sterilized pollen from China that was commonly used as bee feed, and from the ill-advised use of Chinese royal jelly for the priming of queen cells. Both practices appear to have been curtailed, but perhaps a bit too late.
Practical Applications: Well, duh!
Asian bee parasites can also make the jump into the European honey bee when it is introduced (for its superior honey production) into the native territory of the Asian bee. This unnatural contact between the species sets up a scenario in which parasites (including viruses) of the Asian bee can easily transfer to the European bee, as did varroa. Then it is simply a matter of someone illegally smuggling a favored queen, or of a swarm hitching a ride on a ship, from the Orient to any other country.
The honey bees species of Asia host to several viruses: Black Queen Cell , Thai Sacbrood, Deformed Wing, Kashmir, and Apis Iridescent Virus. As yet, it is not clear whether these viruses originated in Asia, or were also native to the European bee. But Jerry Bromenshenk has suggested that perhaps we have simply now imported nearly the entire suite of Asian Bee pathogens to the European bee! (The Tropilaelaps mite of Apis dorsata, and other species of Varroa besides destructor have yet to reach this continent).
Ostfeld notes that “Increasing global trade, which is the root cause of most accidental introductions of alien species, including parasites and pathogens, causes increased connectivity between populations on different continents and in different regions.” As we have seen by the recent rapid worldwide spread of both varroa and N. ceranae, we humans are remarkably efficient at transmitting bee parasites across natural barriers! This sort of connectivity is totally unnatural for honey bees and their pathogens, and just makes it so much harder for the bees and beekeepers.
We are also finding that honey bee parasites are often not too picky about the species that they parasitize, and that honey bee parasites may unwittingly be carried by other species. Elke Genersch (2010), in her excellent review of emerging bee viruses, notes that: “New Zealand native bumblebees are now hosts to a parasitic nematode and three mite species, all of which are thought to have come from the UK with the original introduction of bees. Conversely, in some parts of the world where the European honey bee is an introduced livestock, a still unknown or a benign pathogen of a native species may switch the host and invade the honey bee population with unforeseeable consequences” (like the unforseeable emergence of Deformed Wing Virus as a major bee pathogen).
Each time we inadvertently introduce a new pathogen to the U.S. bee population, the bees must then establish a new equilibrium with their existing suite of pathogens (since 1960 we’ve introduced chalkbrood, tracheal mite, varroa mite, deformed wing virus, small hive beetle, and apparently Nosema ceranae, and likely other viruses). It’s not surprising then, due to the increased parasite load that our bees are carrying, that they are less robust and productive than they used to be, and are perhaps more susceptible to pesticides.
Practical Application: It’s probably a good idea to restrict imports of bees from overseas, and we should encourage Canada and Mexico to do so too. Australia’s rigorous port surveillance and quarantine system should be a model for us.
When studying other host populations to which a novel parasite has been introduced, once the initial phases of epidemic have been resolved, the eventual host population generally reaches an equilibrium at a lower host density than prior to the introduction of the parasite. In the U.S., we may well have enjoyed the glory days of easy beekeeping back when the honey bee would still have been considered to be a relatively new introduced (and invasive) species, before it was burdened with the recent slew of additional pathogens, especially the infection in nearly every single bees these days with Deformed Wing Virus!
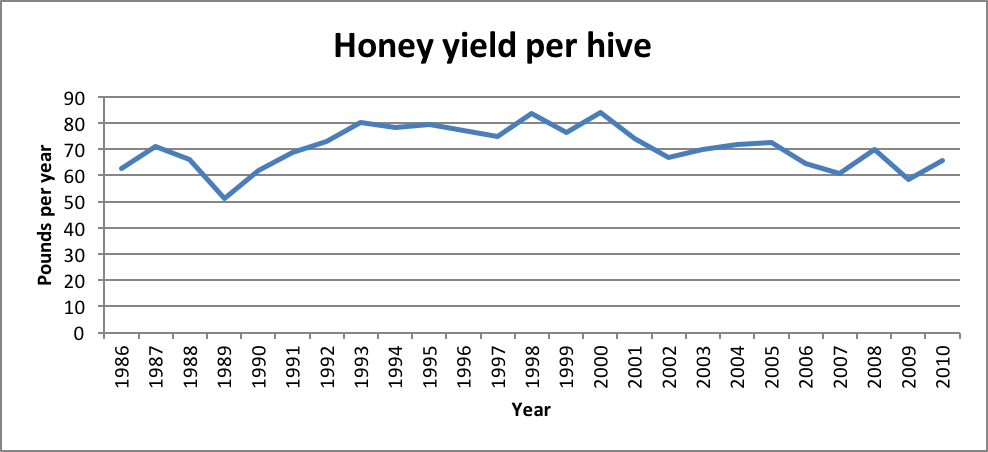
Practical Application: We may not notice that a colony is fighting infections, but the metabolic cost of ramping up the immune response, and the insidious toll on forager lifespan may take a big hit out of colony buildup and honey production (Fig. 2).
About the only good news for the long term is that both host resistance and parasite virulence are respectively evolutionarily costly to both host and parasite, so in the long term, we can likely expect this novel parasite load to eventually become less burdensome to the poor bees.
The Next One
The show is not over! I can guarantee that new strains of bee viruses will either evolve or be introduced into the U.S. Even today there are distinct clusters of Israeli Acute Paralysis Virus circulating about the country (Palacios 2008), some quite virulent. And we may not even recognize that we’ve been hit–Nosema ceranae slipped under the radar; Dr. Joe DeRisi recently reported that our bees are infected with the larvae of a parasitic fly that no one had even noticed! And we should all be scared of the Tropilaelaps mite, the introduction of which would be calamitous to beekeeping in all but the coldest areas of the U.S.
Practical Application: Expect more colony collapse events in your lifetime.
So what will the next plague be, and what will happen when it appears? I found Richard Ostfeld’s 2008 book Infectious Disease Ecology to be quite informative. He notes that nearly half of all the currently emerging disease outbreaks in plants, wildlife, and humans are due to viruses. This is likely due to the ability of viruses to evolve so quickly, and from their ability to jump from host to host. My guess is that most historical outbreaks of bee collapse events were due to the evolution of novel virus strains, coupled with environmental stresses.
Any time that a new pest or pathogen enters a host population, the first wave of infestation or infection may be relatively benign, due to the inefficiency of the pathogen at infecting the novel host. But after a generation or two, the more successful pathogen strains may then explode into a devastating epidemic (remember when tracheal mite first became established the U.S. and killed off about 70% of the colonies?). But then the host fights back, with the survivors displaying a greater degree of resistance to the pathogen, and eventually a more stable host-parasite relationship. Again, this result is evident in the case of tracheal mite, which is now rarely an economic problem in either U.S. or European bee populations (but keep in mind that the only stable host/parasite relationships that we are able to study are those that “worked it out”—we can’t study the ones that went extinct in the process!).
But a steady state is not necessarily the end result. Ostfeld explains that “It is no doubt realistic to acknowledge that many populations spend much of their time far from any equilibrium, recovering from the latest disaster or coming back down to earth after the latest bonanza, and that many [natural] populations are in fact metapopulations, comprising patches that may or may not be occupied at any given time.” This was likely the case with honey bees prior to the widespread domestic management of hives.
Ostfeld also notes that there may be a see-saw balance between immunity to different diseases. For example, in human populations, epidemics of two unrelated diseases, measles (caused by a virus) and whooping cough (bacterial) alternate in a two- to three-year cycle. Even with a single virus, various strains can cause alternating epidemics, as in the case of the mosquito-borne dengue virus, which consists of four related serotypes. Epidemics of each serotype fluctuate out of phase with each other. I wouldn’t be surprised if we find that some bee pathogens follow a similar course.
The bottom line is that we should just get used to the fact that we are going to experience periodic epidemics in our bees, and that by the time anyone figures out what happened, the epidemic may have already run its course, or even possibly set us up for the next one!
Practical Application: Whatever your pathogen problem was last year, it may not be the same next year!
Viruses are Everywhere!
When we see someone coughing and sneezing from a rhinovirus (a cold), or covered with measles rash or chickenpox blisters, we make an effort to avoid being exposed to those contagious viruses. The question then is whether we can avoid exposing our bees to harmful viruses? The answer is: Not likely!
Singh (2010) analyzed 65 honey bee pollen foragers in 2007, and found that all were infected with at least one virus, with most having multiple infections. Honey bees act as their own pathogen vectors, through contact on flowers, through worker and especially drone drift (Brenna Travers found that more than half of all drones may drift to other hives), by the multiple matings of queens with potentially infected drones, and especially through the robbing out of sick and dying hives (surprisingly, the math of evolution apparently finds that the benefit of the stolen honey to be greater than the cost of acquisition of pathogens).
Practical Application: Keep an eye on your yards for failing colonies. Don’t let them get robbed out. If I find a hive that is really sick from something that I don’t recognize, I simply kill the bees and burn the frames. I consider that cheap insurance to keep whatever the heck it was from spreading to the rest of my operation.
Diana Cox-Foster’s team (Shen 2005) detailed the “intricate transmission routes” of bee viruses within the hive via the transfer of jelly from nurse bees, through the eggs of infected queens, and also by being vectored by varroa mites. The nest year, ARS researchers (Chen 2006) confirmed that BQCV, DWV, CBPV, KBV, and SBV could all be passed from queens to their offspring.
Then in 2008, another ARS scientist, Wayne Hunter, found that viruses and bacteria become “aerosolized” when large numbers of bees are defecating in the air. Such infective particles would then be electrostatically attracted to flying bees, likely groomed into their pollen pellets, and then carried back to the hive. One need only imagine the opportunities for such transfer of pathogens in a crowded holding yard!
As if that weren’t enough, when Djikeng (2009) analyzed the waters of a freshwater lake in Maryland, they found four common bee viruses, Cricket Paralysis Virus, and evidence of a previously undescribed novel insect paralysis virus related to the above!
The take home message here is that there is no way that bees can avoid being exposed to viruses! The end result is that every colony is going to “catch” the viruses, but may not exhibit noticeable symptoms. Once the colony manages to mount an effective immune response to keep each specific virus in check, it then becomes an “asymptomatic reservoir” of that virus, sort of like a “Typhoid Mary,” likely contributing to the spread of those viruses to other hives.
Alternative Hosts
The picture is even more complicated than there simply being a reservoir of bee viruses. The reservoir is actually more like a smorgasbord! Remember earlier in this article when I mentioned that a common characteristic of plague-causing pathogens was that they could infect more than one species of host? Well, the “bee” viruses, nosema, chalkbrood, and likely their pathogenic bacteria do just that! And now we’re getting to the real meat of the issue.
A parasite that infects more than one host species must then deal with different resistance mechanisms and life histories (like how quickly the host life cycle is completed). So multi-host parasites may not evolve into the sort of fine-tuned specialists that do little harm to the host, as their mechanisms for gently suppressing the immune system in one host may be devastating to another. Rutrecht (2009) found that some bumblebee species were far more resistant to Nosema bombi than others. Some species simply shrugged off an infection, whereas others were hit hard. Such differences may be involved in the competition between species, with those species that are common and carrying a diverse parasite load having a competitive edge over less common species that are unable to handle the parasites (Durrer 1995). We are currently witnessing the extinction of some species of bumblebees, likely due the “spillover” of introduced parasites from commercial bumblebees, while other species in the same areas continue to thrive (Colla 2008). This would be an example of the creation of a fatal plague in one species simply due to another species acting as a pathogen reservoir.
We think of “honey bee viruses” as being host specific, but in actuality they are rather generic, being also able to infect related bees, wasps, and ants, in some cases varroa mites, and perhaps even flowering plants! A recent eye-opening study by Dr. Diana Cox-Foster’s Penn State team (Singh 2010) detected “bee viruses” in eleven other non-Apis hymenopteran species, ranging from solitary bees to bumble bees and wasps! This finding has profound implications!
In nature, pollinator species compete for the same pollen and nectar resources. If any species starts to dominate the landscape, it is then more likely to suffer from epidemics of viruses or other parasites. This is especially true if the parasites can jump from one host to the other. So this brings up an interesting question: if some of our native bee and wasp species are relatively immune to one of the bee viruses, then they could use that virus as a competitive weapon against competing honey bees by intentionally depositing the virus onto flowers? Cox-Foster’s findings suggest that such bee-to-flower-to-bee virus transmission may be commonplace (Fig. 3):
“For the first time, we report the molecular detection of [bee] viruses … in pollen pellets collected directly from forager bees. Pollen pellets from several uninfected forager bees were detected with virus, indicating that pollen itself may harbor viruses. The viruses in the pollen and honey stored in the hive were demonstrated to be infective, with the queen becoming infected and laying infected eggs after these virus-contaminated foods were given to virus-free colonies.”
They continue:
“Phylogenetic analyses support that these viruses are disseminating freely among the pollinators via the flower pollen itself…Notably, in cases where honey bee apiaries affected by CCD harbored honey bees with Israeli Acute Paralysis virus (IAPV), nearby non-Apis hymenopteran pollinators also had IAPV, while those near apiaries without IAPV did not. In containment greenhouse experiments, IAPV moved from infected honey bees to bumble bees and from infected bumble bees to honey bees within a week, demonstrating that the viruses could be transmitted from one species to another. This study adds to our present understanding of virus epidemiology and may help explain bee disease patterns and pollinator population decline in general.”
Figure 3. Honey bees and other pollinators may swap pathogens on shared flowers. The dynamics of virus interchange among bee and wasp species are complex, and may be involved in the genesis of bee plagues. Photo by Sarah Greenleaf.
The authors have suggestive evidence that the viruses may actually be inside the pollen, implying that the viruses might replicate in the plants themselves! This is not unheard of, as there is evidence of related viruses transmitting similarly between plants and aphids.
Practical Application: More than 200 tons of honeybee-collected pollen is used annually for bumble bee rearing worldwide. This pollen, unless irradiated, is a likely vector for insect and plant viruses worldwide. It is unwise to feed non-irradiated pollen!
They also found that DWV could be transmitted from beebread to the queen (presumably via infected jelly produced by nurse bees), and that the newly-infected queen would begin laying infected eggs. The beebread was infective even after six month’s storage! Surprisingly, it also appeared that the queen could selectively clear infection by Sacbrood while continuing to be infected by DWV!
Practical Application: Cox-Foster’s findings confirm that viruses can remain infective in the beebread in deadouts. This may help to explain why the irradiation of deadout equipment appears to allow restocked packages to build up better. Unfortunately, short of radiation, there are no antiviral treatments that I know of that will penetrate the beebread.
The implications of Cox-Foster’s findings are enormous in helping to understand why bees suffer sporadic plague epidemics (I’ve been leading up to this for some time). Remember the concept of viral quasispecies—that a virus exists as a “cloud” of slightly different mutated forms? Schneider (2001) explains:
“The theoretical advantage of maintaining a diverse quasispecies is that, when the virus is shifted to a new environmental niche or selective regimen, a variant may already be present in the population which will be more fit in the new environment.”
But what happens when a virus jumps from one host to another, which will likely strongly favor one variant over the rest?
“However, excessive diversity can create problems if the virus is subjected to repeated bottlenecks. Since most mutations are deleterious, frequent bottlenecks can result in the rapid loss of fitness known as Muller’s ratchet. In order to survive, a virus must be diverse enough to adapt rapidly to changing environments without losing fitness during passage from host to host.”
Schneider performed an elegant experiment in which he tracked the genetic diversity of the viral cloud during serial passages of two plant viruses in single host species, and then tracked what happened when he introduced a single clone (one specific variant) of each virus into new host species. Amazingly, the virus clones rapidly replicated (doubling about once and hour), and generated enough mutations to recreate a cloud of variants roughly equal to that of the original host cloud! But the new cloud was different from the original—it was now adapted to the new host.
But different hosts put different pressures on the viruses and surprisingly, on their mutation rates:
“This suggests the possibility that different hosts may accelerate or decelerate the rate of viral evolution by permitting or denying high levels of diversity in viral populations. Diversity in viral quasispecies has been described previously as a mechanism to avoid host resistance responses or a reservoir to maintain variants with selective advantages in other environments and has been correlated with the ability to infect numerous hosts.”
And the key issue here is:
“Perhaps, the very resistance mechanisms that [the alternative hosts] use to combat viruses are in fact generating high-diversity quasispecies that act as a source of new pathogenic variants.”
This point is hammered home by Agudelo-Romero (2008):
“Sometimes, a virus may spill over from its usual host species into a novel one, where it usually will fail to successfully infect and further transmit to the new host. However, in some cases, the virus transmits and persists after fixing beneficial mutations that allow for a better exploitation of the new host. This situation would represent a case for a new emerging virus” [emphasis mine].
And there we have it folks! As bee pathogens jump from one host to another, the evolutionary pressure of adapting to different host resistance mechanisms is likely to create new forms of viruses that may be more virulent when they hop back to the original host. In nature, everything is connected to everything else!
Areas rich in pollinator species may be evolutionary “hot spots” for parasite evolution. Dupas (2003) explains: “Variations observed in parasite virulence and host resistance may be the outcome of coevolutionary processes. Recent theoretical developments have led to a ‘geographic mosaic theory’ of coevolution according to which there are some localities where reciprocal selection occurs (hot spots) and others where it is strongly reduced (cold spots).”
This theory suggests that in areas in which bees interact with numerous species of other pollinators, there would be more chance of novel virulent forms of pathogens to develop. Luckily, research by the same author (Dupas 1999) suggests that there is an adaptive cost to a virus in order to maintain genes for strong immunosuppression of the host, and that in areas where the virus infects multiple hosts, that that cost constrains the development of highly virulent forms.
How Do the Bees Keep Up?
So how do bees deal with this constantly-changing weaponry of their parasites? Largely by sex! Sexual reproduction is nature’s way of creating genetic diversity in order to keep ahead of parasites (Lively 1996).
The most conducive set up for an epidemic is when all the potential hosts are closely related. We’ve seen this again and again in agricultural monocultures, such as with the devastating epidemics that decimated clones of potatoes in Ireland, bananas in Central America, and coconuts in the Caribbean—all those plants were propagated as identical clones, and when a virulent pathogen came along to which that clone was susceptible, it roared through those artificial populations like wildfire!
In order to prevent such epidemics in field crops, farmers routinely plant mixes of varieties seed—with a proportion of them being resistant to different pests (Zhu 2000). The genetic mixture slows the propagation of the parasite enough to prevent a plague from getting underway. Exactly the same effect occurs when bee colonies consist of a genetic mixture of workers due to the multiple mating of the queen, and when the overall population is genetically diverse (Whitehorn 2011).
Practical Application: Mix it up in large operations. Avoid having all colonies in a yard coming from the same mother. If you raise your own queens, make sure that they get to mate with a diversity of drones.
The Four Horsemen
I suggested in a previous article that beekeepers should always be alert for “The Four Horsemen of Bee Apocalypse”: Famine (the lack of adequate pollen or winter stores), cold (especially spring or fall snap chills), pestilence (parasite buildup), and toxins (either natural or manmade). Any combination of two or more of the above can initiate a chain of events that can lead to the rapid depopulation of colonies (see Sick Bees 2). Bee plagues are generally associated with one or more of the Four Horsemen.
Practical Application: Most incidences of epidemic collapses in apiaries can be traced back to the presence of one or more of the Horsemen. The beekeeper, by intervening, may be able to prevent the situation from spiraling into a collapse event.
Bees as (Semi-) Domestic Animals
In reference to commercial beekeeping Dr. Gordon Wardell explains that “We are trying to shoehorn a wild animal into an intense agricultural management system.” I’m typing this article shortly after almond pollination. Talk about exposing the bees to the Four Horsemen! Prior to, and just after bloom, the poor bees suffer from famine (since there is virtually nothing to eat on the orchard floor), from unexpected cold snaps, are exposed to strains of parasites from all corners of the country, and must detoxify both the natural amygdalin in the almond pollen, as well as the fungicides and insect growth regulators from the incessant spraying of the bloom (Fig. 4)!
If the almond bloom weren’t so nutritious and stimulatory to broodrearing, many colonies would not be able to recover from the pre bloom stress. And some in fact don’t. And if they are not moved out of the orchards soon enough after the petals fall, strong colonies can quickly get sick due to lack of pollen income.
Practical Application: The midwinter period just prior to almond bloom, when beekeepers are trying to stimulate hives sitting in forage-poor holding yards, is stressful to the bees, and the situation is ripe for a pathogen epidemic to take off.
Figure 4. This forager is packing on almond pollen. She is unaware that a spray rig passed by minutes before, blasting that pollen with a tank mix of a fungicide and an insect growth regulator. Almond pollen, though highly nutritious to bees, is often heavily contaminated with fungicides. Photo by the author.
In nature, plagues are largely self- limiting. As a virulent parasite kills off the host, eventually a point is reached where there are no longer enough uninfected nearby hosts for the epidemic to propagate, and it simply burns itself out. Any survivors then find that there is no longer competition from the deceased and therefore more food is available, which then helps the population to recover, founded by the genetics of the survivors.
In commercial agriculture, this sort of self limitation on plagues does not take place. When one makes their living with bees, the financial constraints of the economy of scale butt head-on into the biological reality of infectious disease epidemiology.
In natural populations, virulent virus strains often simply go extinct, as they are unable to maintain an infective reservoir in the normal scattered and genetically diverse host population. Unfortunately, large-scale beekeeping (which would include even high colony densities due to hobby beekeepers) tends to create monocultures of closely-related bees, which are continually replenished should they perish to a parasite, thus providing enough new fodder to perpetuate a plague that would have burnt itself out under more natural conditions.
Brown and Fries (2008) explain the situation in large operations:
“In natural populations, where opportunities for horizontal transmission are likely to be low, such an epidemic [of a virulent virus strain] might rage through a colony, resulting in colony death, but would be unlikely to spread through the honey bee population (that is, it would be an intra- rather than inter-colony epidemic). However, in the managed system where opportunities for horizontal transmission are high, such an epidemic might well run through the larger honey bee population, causing mass colony mortality before dying down. Thus, at the population-level, viral impact may simply be a phenomenon of honey bee management.”
Practical Application: Manage each yard of bees as its own population, and be careful about mixing sick yards with healthy ones.
Epidemics used to also be limited by geographical barriers, such as mountains and deserts. Nowadays, however, we tend to homogenize all the pathogens in the country every year. Roughly half of all managed hives are moved to California’s almond orchards each winter. And then, nearly half of all honey-producing hives in the country are taken to only three states–California and the Dakotas.
Practical Application: Look for isolated locations and watch out for who you set down next to!
Colony resistance to pathogens is largely a function of good nutrition, which is often not to be had when bees have only agricultural monocultures to forage upon. That, coupled with the inordinate amount of pesticides used on crops often leaves colonies standing on weak immunological legs. Some commercial beekeepers are finding that it is simply not worth that extra pollination contract during the summer, and that their hives look better going into winter if they get a break from the toxin-laden agricultural lands.
Practical Application: A number of beekeepers have found that it is worthwhile to reduce the numbers of colonies per yard. The overall return per yard may actually be greater with fewer colonies.
In fall and spring holding yards, many bee operations resemble CAFO’s (Concentrated Animal Feedlot Operations). Again, as with other domestic livestock, when one unnaturally crowds animals like this, one creates perfect breeding grounds for the next epidemic. Poultry CAFO’s are sealed against outside birds, which might bring in a pathogen; unfortunately, there’s no way to isolate a bee operation from other managed or native bees. Other animal operations routinely treat their stock with antibiotics and pesticides to prevent epidemics of bacteria, lice, or mites. Large-scale beekeepers do the same. We should be aware, though, that we know little about how such treatments affect the critical beneficial microflora in the bee guts and beebread, or the bees’ immune response to viruses. It surprises me that some beekeepers throw every treatment in the book into their hives each year—something that they would never consider doing to their own children!
Practical Application: In large-scale operations, one should be judicious about the cost/benefit analysis of prophylactic treatments. Such treatments not only cost money, but confer metabolic costs on the bees, and may disrupt the balance of beneficial endosymbionts that normally suppress viruses, AFB, nosema, and chalkbrood. It would be wise to practice Integrated Pest Management, and only apply treatments if pest monitoring indicates that there is actually a problem brewing.
Brown and Fries go on to say:
“Given the potential for rapid mutation and mutant swarms in RNA viruses, it seems likely that these viruses have already evolved away from their pre-mite state. Because beekeepers remove the selective disadvantage of being virulent at colony level, by removing the vector through mite control measures, the current problems with virus induced colony mortality are likely to continue, or even increase, unless mite tolerant stock and/or virus resistant stock is developed. As demonstrated in natural systems, although mite infested colonies are likely to succumb to virus infections if left untreated, the species A. mellifera is unlikely to perish without the involvement of apiculturists” [emphasis mine].
Practical Application: Allow me to quote cartoonist Walt Kelly: “We are confronted with insurmountable opportunities.”
It’s up to us, as beekeepers, to work with nature to resolve the problem that we accidentally created by introducing the varroa mite to the European honey bee. The solution is to breed for resistant bee stocks so that we can wean our bees back off of chemical support. It’s not going to happen overnight, but great progress has been made in the past decade (my hat is off to Drs. Hoopingarner, Harbo, Danka, Harris, Villa, and especially Rinderer for their foresight and hard work). In my own operation, I find that by breeding for naturally-resistant stock, that my bees look better, I have no need for synthetic miticides, rarely use any antibiotic, and most importantly, make more money!
Coming to Terms with Colony Collapse
I’m going to let Keith Delaplane (quoted by Reese 2011), summarizing the accomplishments of the Coordinated Action Project for CCD, summarize the situation regarding our latest plague–CCD:
“One of our biggest frustrations has been defusing the expectation for ‘a cure’ for CCD. The answer, when it comes, will be a knowledge-based enterprise, not a product-based enterprise. The answer will be messy.
“The answer will include management decisions like lower-density apiaries, integrated pest management adoption, selective breeding programs, as well as new technologies like RNAi and marker-assisted breeding.
“Nothing about this is easy. Bee decline is a systemic problem on a continental scale. But you know what; we’ve shortened the list, made important discoveries in toxicology and disease and are coming up with practical answers in disease remediation and genetic resistance.”
Meanwhile, many beekeepers are simply practicing good commonsense animal husbandry with their bees, and being very successful at it!
References
More information on the CCD Coordinated Action Project (CAP) can be found at (Broken Link!) http://www.beeccdcap.uga.edu/
Achtman, M (2007) Microevolution and history of the plague bacillus, Yersinia pestis. PNAS 101(51): 17837-17842.
Agudelo-Romero P, P Carbonell, MA Perez-Amador, SF Elena (2008) Virus adaptation by manipulation of host’s gene expression. PLoS ONE 3(6): e2397.
Brown, MJF and I Fries (2008) Evolutionary epidemiology of virus infections in honey bees. In Virology and the Honey Bee.
Chen, YP, JS Pettis, A Collins, MF Feldlaufer (2006) Prevalence and transmission of honeybee viruses. Applied and Environmental Microbiology 72(1): 606-611.
Colla, SR and L Packe (2008) Evidence for decline in eastern North American bumblebees (Hymenoptera: Apidae), with special focus on Bombus affinis Cresson. Biodivers Conserv 17:1379–139
Djikeng A, R Kuzmickas, NG Anderson, DJ Spiro (2009) Metagenomic analysis of RNA viruses in a fresh water lake. PLoS ONE 4(9): e7264. doi:10.1371/journal.pone.0007264
Dobson, A, and J Foufopoulos (2001) Emerging infectious pathogens of wildlife. Phil. Trans. R. Soc. Lond. 356: 1001-1012. http://rstb.royalsocietypublishing.org/content/356/1411/1001.full.pdf
Dupas S and M Boscaro (1999) Geographic variation and evolution of immunosuppressive genes in a Drosophila parasitoid. Ecography 22: 284–291.
Dupas S, Y Carton, M Poirie (2003) The genetic dimension of the coevolution of virulence resistance in Drosophila-parasitoid wasps relationships. Heredity 90:84–89.
Durrer, S and P Schmidt-Hemple (1995) Parasites and the regional distribution of bumblebee species. Ecography 18:114-122.
Genersch, E and M Aubert (2010) Emerging and re-emerging viruses of the honey bee (Apis mellifera L.). Vet. Res. 41:54 http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2883145/
Genersch, E and M Aubert (2010) Emerging and re-emerging viruses of the honey bee (Apis mellifera L.). Vet. Res. 41:54.
Hunter, W.B., CL McKenzie, BW Mitchell (2008) Collecting viruses from the air. The 72nd Annual Meeting of the Florida Academy of Sciences.
Lively, CM (1996) Host-Parasite Coevolution and Sex. BioScience 46(2): 107-114. http://www.indiana.edu/~curtweb/L567/readings/Lively1996.pdf
Meana, A, M Higes, R Martín-Hernández (2010) Nosema ceranae in Europe: an emergent type C nosemosis. Apidologie (41) 3: 375-392.
Palacios, G.P., et al (2008) Genetic analysis of Israeli acute paralysis virus: distinct clusters are circulating in the United States, Journal Virology 82: 6209-6217.
Reese, A (2011) Honing in on bee colony collapse disorder. Western Farm Press, Feb. 17, 2011.
Rutrecht, ST and MJF Brown (2009) Differential virulence in a multiple-host parasite of bumble bees: resolving the paradox of parasite survival? Oikos 118, 941–949. (doi:10.1111/j.1600-0706.2009.
Schneider WL and MJ Roossinck* (2001) Genetic diversity in RNA virus quasispecies is controlled by host-virus interactions. Journal of Virology 75(14): 6566-6571http://jvi.asm.org/cgi/reprint/75/14/6566.pdf
Shen, M, L Cui, N Ostiguy, D Cox-Foster (2005) Intricate transmission routes and interactions between picorna-like viruses (Kashmir bee virus and sacbrood virus) with the honeybee host and the parasitic varroa mite. J. Gen. Virol.86: 2281 – 2289.
Singh R, AL Levitt, EG Rajotte, EC Holmes, N Ostiguy, et al. (2010) RNA viruses in hymenopteran pollinators: evidence of inter-taxa virus transmission via pollen and potential impact on non-Apis hymenopteran species. PLoS ONE 5(12): e14357. doi:10.1371/journal.pone.0014357
Whitehorn, PR, et al (2011) Genetic diversity, parasite prevalence and immunity in wild bumblebees. Proc. R. Soc. B 278(1709): 1195-1202.
Zhu, Y (2000) Genetic diversity and disease control in rice. Nature 406: 718-722.