When To Feed Pollen Sub
September 17, 2015
When To Feed Pollen Sub
Randy Oliver
ScientificBeekeeping.com
First published in ABJ in November 2014
Introduction
Beekeepers often ask me at what time of the season they “should” [1] feed colonies pollen sub. My recent trial of pollen subs demonstrated the clear benefit of feeding patties of either natural pollen or artificial pollen substitutes in time of pollen dearth. However, it also indicated that there was no apparent benefit once natural alder pollen became abundant in January. I was thus curious as to whether this held true later in the spring. In order to answer that question, I decided to run a follow up trial.
Materials, Methods, and Trial Log
We ran the trial in a nonagricultural setting the Sierra Foothills during our spring nectar and pollen flow. For the test yard we set up 30 5-frame nucs transferred into deep singles, placed in pairs, with entrances splayed in different directions. All were headed by recently-mated queens from the same mother (mated in their nucs), all exhibiting good brood patterns. Over a period of ten days in early May we carefully equalized the colonies for strength. We swapped combs as necessary so that each hive contained three frames of brood (typ. 2 sealed, 1 open); one frame of honey/pollen and one drawn comb. We then swapped colony positions or added shook bees to equalize colony strength such that there was complete coverage of the outer combs with bees (Fig. 1).
 Figure 1. We began the trial by equalizing all colonies to three frames of brood and a total of five frames covered with bees, adding shook bees, if necessary, brought to the yard in packages.
Figure 1. We began the trial by equalizing all colonies to three frames of brood and a total of five frames covered with bees, adding shook bees, if necessary, brought to the yard in packages.
During the equalization process, we confirmed that all the colonies were free of signs of disease, and for good measure applied a drench of ½ cup 1:1 sugar syrup containing 95 mg fumagillin and 100 mg OTC, as preventative treatment for nosema and EFB.
Upon completion of equalization, we filled each hive to 10 frames total by adding 5 frames of extracted drawn combs, one dark and one light comb immediately on either side of the nuc, filling the rest with an “average” assortment of combs, distributing combs from each donor super to several different hives in order to minimize any effect of combs from any particular super. We then added a 1½-inch feeder rim.
May 24: Day Zero, Patty feeding #1 All colonies at exactly 5-frames strength, with equal amounts of brood. The natural main nectar/pollen flow had recently begun, with a wide variety of plants in bloom, mainly Himalayan Blackberry (Fig. 2)—a known source of high-quality pollen. We randomly assigned one hive in each pair to be given pollen sub patties (Treatment group); the other served as a Control.
We fed all Treatment colonies one 1-lb pollen sub patty [2]. In order to control for the effect of the sugar contained in the pollen sub (48% by weight), we made up sham patties containing an identical amount of sugar alone [3].
 Figure 2. A view through the bear fence on June 3rd. You can see the blackberries in bloom in the background, and plantain in bloom in the foreground. There was a good diversity of natural pollen sources throughout the course of the trial.
Figure 2. A view through the bear fence on June 3rd. You can see the blackberries in bloom in the background, and plantain in bloom in the foreground. There was a good diversity of natural pollen sources throughout the course of the trial.
June 3: Blackberry flow coming on, with plenty of nectar shaking from the combs [4]. Spot checked hives for patty consumption. The Treatment hives had consumed only a small amount (Fig. 3); all checked hives had completely devoured every speck of the fondant in the Control patties.
 Figure 3. Note the minimal in consumption of the pollen sub patty (foreground) after 10 days, whereas the bees had devoured every bit of fondant from the control patty in the background. Such avid consumption of the fondant was typical throughout the trial, despite abundant nectar coming in.
Figure 3. Note the minimal in consumption of the pollen sub patty (foreground) after 10 days, whereas the bees had devoured every bit of fondant from the control patty in the background. Such avid consumption of the fondant was typical throughout the trial, despite abundant nectar coming in.
Questions raised: We knew from previous experience that the pollen sub we used is normally extremely attractive to bees, and readily consumed. Yet in this trial the bees ravenously consumed the fondant patties, and initially ignored the pollen sub patties, despite their being half composed of sugar. Were the bees simply not interested in additional protein when natural pollen is plentiful? The second question that occurred to us was whether we should consider feeding fondant to growing nucs in spring even during a nectar flow?
June 10: Syrup feeding #1. Acting on the bees’ eagerness to consume sugar despite the nectar flow, we fed ½ gal of 1:1 sucrose syrup to encourage build up. Spot checked for patty consumption—some colonies had nearly finished their patties. June 12 Patty feeding #2. Most syrup consumed, fed sub and sham patties.
June 17: Plenty of blackberry buds remaining on plants. All sugar patties fully consumed by 5 days. Pollen sub patties partially consumed.
Observation: We noted residues of pollen sub at the entrances of most Treatment hives, suggesting that the bees might be “consuming” them for the sugar only, and discarding the expensive protein.
June 26: Patty feeding #3 Pollen sub patties nearly completely consumed in most hives. Hives becoming well filled, comb building in rim space, need to add supers. Plenty of mixed bloom (Fig. 4), some blackberry bud remaining, Yellow Star Thistle coming on.
 Figure 4. Throughout the trial we spot checked to verify the presence and diversity of beebread in the combs. In this photo from June 26, the beebread consisted mainly of gray blackberry pollen, but other colors were also present, indicating a healthy diversity of nutrition.
Figure 4. Throughout the trial we spot checked to verify the presence and diversity of beebread in the combs. In this photo from June 26, the beebread consisted mainly of gray blackberry pollen, but other colors were also present, indicating a healthy diversity of nutrition.
July 2: Time Point 1 Graded all colonies for strength [5]—all approaching filling their boxes. Will need to super up soon! Colonies had not yet consumed their pollen sub patties.
July 7: Syrup feeding #2 Nectar flow beginning to taper off, but had waited a bit too long to super up (due to lack of equipment), so many rims were filled with honeycomb. Scraped it out and added a deep of foundation. Some colonies had recently finished consuming their sub patties (the sugar patties were long gone), so plan to feed more tomorrow. Spot checked for presence of beebread. Adequate ring around the broodnests, at least 4 colors of pollen. Fed all ½ gal 1:1 sucrose syrup. July 8 Patty feeding #4. Added 1 deep super of foundation.
July 21: Syrup feeding #3 Fed ½ gal 1:1 sucrose syrup. Spot checked several—most, but not all, had drawn a few frames of foundation in the second box. Very little beebread in the combs—time to end the trial.
July 22: End Point Final grading of colonies for strength. We censored the data from one Treatment colony in which the queen had been superseded.
Results
Despite the fact that many colonies had tripled in strength, there was no difference in colony buildup between the two treatment groups (Fig. 5). There was also surprisingly little colony-to-colony variation in buildup. The range of final strength grades for the Control group was 11-16 frames of bees; for the Treatment group 12-16 frames.
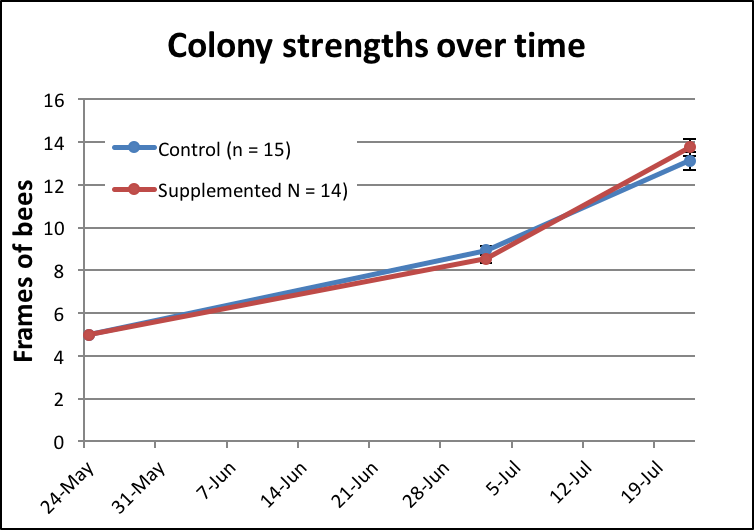 Figure 5. There was no statistical difference in buildup between the two groups (Mann-Whitney p = 0.38). Error bars (SEM) are indicated, but there was so little variation that they hardly show.
Figure 5. There was no statistical difference in buildup between the two groups (Mann-Whitney p = 0.38). Error bars (SEM) are indicated, but there was so little variation that they hardly show.
Discussion and Acknowledgements
Practical application: there was no apparent benefit to feeding pollen sub when there was adequate pollen of high nutritive value naturally available. So don’t waste your money.
Indeed, it appeared that the colonies rather ignored the patties, and may even have licked the sugar out of them and discarded some of the protein. On the other hand, it surprised us how voraciously the colonies chewed through the waxed paper and consumed the sugar fondant, despite there being an abundance of fresh nectar in the combs.
It is important to note, however, that not all natural pollen is of high nutritive value, and that as I pointed out in the large-scale trial, colonies may even collect and make beebread from fungal rust spores or other material, which might even be worse than nothing at all. In such cases, it would then be of great benefit to feed pollen sub.
Practical application: just because there is beebread around the brood, that does not necessarily mean that the colony is enjoying good nutritional intake.
So how can you tell? When “should” you feed?
Practical application: don’t ask what you “should” do—learn to read the combs yourself. The bees will tell you!
How To Tell When Colonies Have Adequate Protein
Open some hives and pull frames out of the broodnest and answer these questions:
- Are the colonies rearing open brood (larvae) in earnest? During protein shortage the nurses will cannibalize eggs and young larvae.
- Do the larvae have a high rate of survival to pupation, as indicated by solid patterns of even-aged larvae making it to pupation (capping) (Fig. 6). Incomplete nutrition will result in poor larval survival or disease.
 Figure 6. Note the solid pattern and even ages of the maturing larvae to the left, indicating a high rate of survival.
Figure 6. Note the solid pattern and even ages of the maturing larvae to the left, indicating a high rate of survival.
- The most reliable indicator (the main one that we use) is to look closely at the very young larvae to see the amount of jelly that the bees are placing around them (see Figs. 7 & 8).

Figure 7. When a colony is under nutritional stress, the nurses cut back on the amount of jelly that they place around the young larvae, as above. I call this “dry brood.”
Practical application: colonies exhibiting dry brood will typically go downhill quickly, or succumb to disease.
 Figure 8. On the other hand, nurses enjoying good nutrition will flood the young larvae with jelly. I call this “wet brood.” I took this photo during a drought-induced pollen dearth in September, after dry-feeding the yard a pollen sub for 10 days. The nurses responded favorably, as evidenced by the larvae now “swimmin’ in jelly.”
Figure 8. On the other hand, nurses enjoying good nutrition will flood the young larvae with jelly. I call this “wet brood.” I took this photo during a drought-induced pollen dearth in September, after dry-feeding the yard a pollen sub for 10 days. The nurses responded favorably, as evidenced by the larvae now “swimmin’ in jelly.”
Practical application: colonies with brood that looks like the above are thriving, and are better able to deal with parasites, pathogens, and pesticides.
In my own operation in the bone-dry Sierra foothills, I have been able to eliminate my summer migration to better forage by learning to read the brood. We immediately start feeding pollen sub (and perhaps stimulatory sugar syrup) as soon as we see the nurses starting to ration jelly during the late summer pollen dearth. Although I much prefer natural forage (and the fact that it’s cheaper for me to move bees to better pasture than it is to feed them), I also weigh the carbon impact of trucking, and the lifestyle cost of being on the road so much.
I will continue this series with the results of additional research that we’ve conducted on the beekeepers’ buck.
Acknowledgements
I’d like to thank my sons Eric and Ian, and helpers Brion Dunbar, Richard Terrel, and Lori Codey. This research was funded by beekeeper donations to ScientificBeekeeping.com, and a grant from the North Dakota Department of Agriculture via the North Dakota Beekeepers Association.
Footnotes
[1] In my book, there are few “shoulds” in beekeeping. Nearly every beekeeping decision involves a complicated cost/benefit analysis involving one’s goals, realistic options, preferences, financial considerations, ability, and values.
[2] We used factory-prepared patties of one of the top-scoring pollen subs in my previous trial.
[3] In order to eliminate the sugar in the pollen sub patties as an added variable, we prepared sham sugar patties for the Control group by kneading a small amount of 50:50 sucrose/HFCS blend into Drivert sugar to the consistency of fondant, and then weighed and rolled them between thin waxed paper “pick up sheets” of approximately the same size as the paper sheets of the pollen sub patties. For the remainder of the trial, each group received matching amounts of sugar, each providing approx. 0.48 lbs of sugar with each patty feeding.
[4] For those unfamiliar with the term “nectar shake” or “shaking nectar,” that means that we could easily shake fresh nectar from withdrawn combs, indicating that a strong nectar flow was on.
[5] All colonies were quickly graded in early morning, with me as sole grader, before the clusters broke, by tipping the boxes back and counting the number of interstices filled with bees.




