Powdered Sugar Dusting – Sweet and Safe, but Does it Really Work? Part 3
Powdered Sugar Dusting—Sweet and Safe, but Does it Really Work?
Part 3
Randy Oliver
ScientificBeekeeping.com
First Published in ABJ in Feb. 2009
Update 2016: although sugar dusting can indeed cause a substantial proportion of the phoretic mites to drop off the bees, in order to effectively manage varroa, dusting requires more effort and repetition than most beekeepers are willing to devote to their hives. In my own operation, we now manage varroa with splitting, drone trapping once a season, and the use of formic acid, thymol, and oxalic acid, plus selection of queens from colonies that exhibit the least buildup of mites.
Scientific inquiry can be exciting yet exasperating, satisfying yet frustrating! On these pages I share my own voyage of discovery, and enjoy the liberty of avoiding scientific formality. If one were to only read published papers, one might think that every experiment worked the first try, successfully answered a question, or made some meaningful discovery. In reality, however, research frequently does not go as planned, and the results may not be at all what you expected or hoped for. The best you can then do is to chalk it up to more experience under your belt, and to see if you can be clever enough to extract any useful information from the data.
In my previous two articles I reported on a field trial of powdered sugar dusting as a mite control method, then on another experiment to determine the rate of mite drop after dusting, and finally at the correlations between the natural mite fall, the infestation level as determined by an alcohol wash of adult bees, and the accelerated mite drop after sugar dusting.
Everything was looking just peachy until I tried to make two figures jibe: the percent mite fall after dusting just wasn’t matching up with the 30-50% figure that I found when I sacrificed a few small colonies, as described previously. So I bit the bullet and ran yet another dusting test!
On a warm afternoon this October, we tested sugar dusting on nine, 10-frame doubles, each containing from 9 – 20 frames of bees. We split each hive open, graded it for number of frames covered with bees, then vacuumed an approximate 300 – 500 bee sample from each hive for an alcohol wash. We then top dusted each hive, caught the mite drop on a dry board for exactly an hour, and then took yet another sample (Fig.1) for a second alcohol wash. We dusted the hives at “production speed”—that is we did not take special effort to dust carefully. Note that our screened bottom boards have a 1-3/8” rim inside the beeways, so some sugar and mites may have remained inside the colonies.
Figure 1. Vacuuming up a bee sample for an alcohol wash to determine the level of mite infestation. Samples were taken from the top bars of the brood nest. Post dusting samples were taken of “average dusted” bees from the same area.
Back home we washed the mites from the bees (Fig. 2) then “counted” the bees volumetrically (using the factor of 3 damp bees per 1ml of volume when shaken down). From these figures we hoped to confirm the actual efficacy of sugar dusting by comparing the pre-dusting infestation level to the post-dusting level (once again hoping that bee science could be that simple). Also, by multiplying the initial infestation level by the number of frames of bees, and then by the factor of a conservative 3000 bees per frame, we could extrapolate the total colony phoretic mite population, and compare it to the 1-hour dusting drop. The results are shown in Table 1.
Figure 2. My son and partner, Ian, washing mites from bees in an alcohol bath.
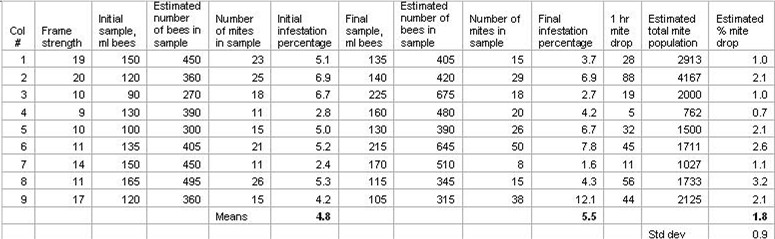
Table 1. Data from dusting 9 colonies, with before and after alcohol wash mite counts, and an estimation of the total phoretic mite population for each colony (initial infestation x number of frames x 3000 bees per frame).
The main assumption of this test is that an alcohol wash of 300 -400 bees from the brood nest would be a consistent and accurate assessment of the mite infestation level. There was actually little reason to assume this, since Fuchs (1985) found that estimating the mite infestation level from a sample of washed bees was rather inaccurate, and could vary by a factor of three up or down! In addition, the response of a colony to dusting is for the bees to move away from the brood area, which may also have affected our data.
Not surprisingly, we found that a single alcohol wash sample of bees is not necessarily a good indicator of mite infestation level–the infestation percentage came back higher after dusting in four of the nine colonies, despite the fact that mites had been removed from the colony by dusting!
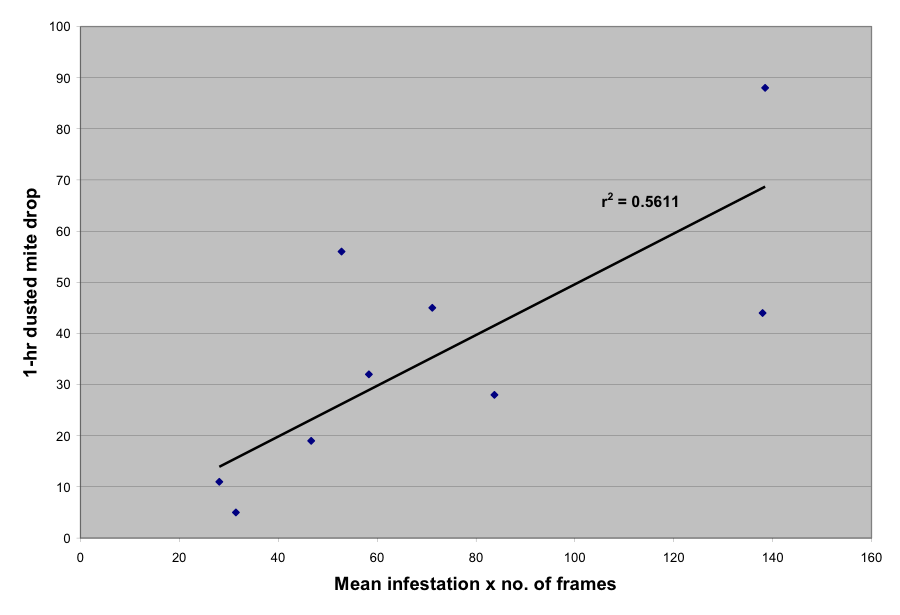
So to try to salvage some meaningful data from this exercise, let’s see how well the sugar dusting drop correlated with the (approximate) mite infestation level as determined by the means of two alcohol wash samples. In Figure 3 I plotted the 1-hr dusted mite drop vs. the mean (of the first and second readings for each colony) mite infestation multiplied by the number of frames of bees in each colony (e.g., a 10-frame colony with a 5% infestation would have a score of 50 on the x axis. You’ll notice that there is a decent correlation. In other words, the dusted mite drop appears to be a quick and fairly accurate method for assessing a colony’s mite level.
Figure 3. Plot of 1-hour mite drop vs. total colony mite population, estimated by multiplying the number of frames in a colony by the percentage infestation determined by alcohol wash (10 frames of bees at 5% infestation would be placed at 50 on the x axis).
By doing a bit more math to adjust for the 10-minute drop that I personally use in the field (see Figure 5 in my previous article) the data confirms that if I want to keep my mite populations below a 1% infestation of the adult bees, that for a 14-frame colony, I should only see 3 – 4 mites on the dry board ten minutes after dusting. There would be proportionally more mites for a 20-framer (5 mites), and of course fewer for a weaker colony. (This infestation level would correspond to an ether roll of 3 mites in a 300-bee sample).
Let me now address my original question: what percentage of the total phoretic mite population actually dropped in the hour after dusting? Well, if we go by the premise that the mites are evenly distributed over the bees (this is likely not true, as Fuchs found that they tend to hang out on bees on the brood frames), and that there were about 3000 bees per frame, then we only dropped less than 2% in the first hour. I’ve already demonstrated that the first hour drop constitutes roughly half of the total induced drop, so the data from this tiny test indicate that there was only a 4% drop overall. This is a far cry from the 30 – 50% drops that I confirmed when I actually sacrificed whole colonies! Another observation that makes me question this low figure is that some colonies in the 24-hour test dropped over 400 mites (doubles) and 800 mites (singles). If the 4% figure were correct, that would imply that one of the (apparently healthy) singles contained over 20,000 mites—a questionable figure, since its 24-hr natural fall of 104 mites indicated a much lower population.
So what gives? Honestly, I don’t know. The whole colonies that I had sacrificed previously were heavily infested and on the small side (sacrificial “goners”), and perhaps dusting was more effective than in large colonies. I’ve noted that sugar dusting may be less effective when the frames are not aligned in doubles, but this only occurred in about a third of the colonies in this test. My guess is that the problem was in extrapolating the results of an alcohol wash of a small bee sample from the brood nest to the entire colony population.
Clearly, what needs to be done to answer this question definitively is to dust and sacrifice more full-sized colonies. Unfortunately, this is an expensive proposition for a cash-strapped beekeeper depending upon almond pollination for his income, so I hope that someone else steps up to the plate to do it!
The take-home message is that the results of this series of tests lead me to question the reliability of either natural mite fall or the alcohol wash (or ether roll) as monitors of mite infestation level! It appears to me that a whole-colony mite drop accelerated by sugar dust (or other mite dislodging agent) is likely the most accurate field-practical way to determine a colony’s mite level.
Update July 7, 2015: We have not used sugar dusting for some years, since we did not find it to be cost effective for us (thymol, formic acid, and oxalic acid gave us more cost effective mite kill). However, since I wrote these articles, I’ve gained some proficiency in modeling bee and mite population curves.
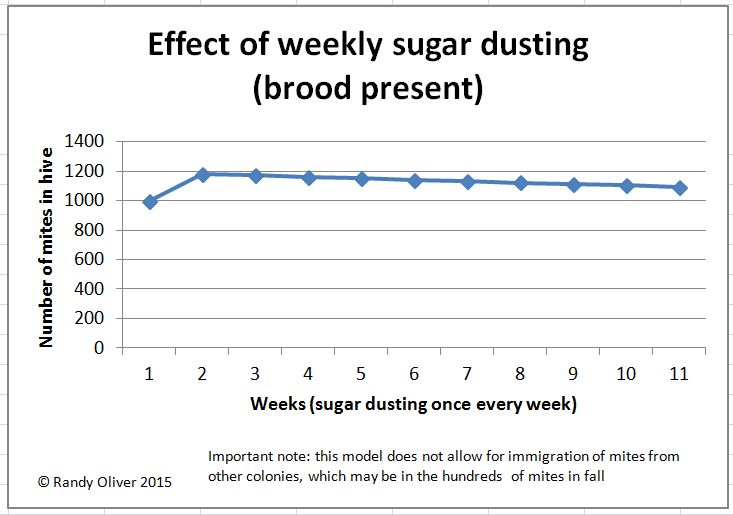
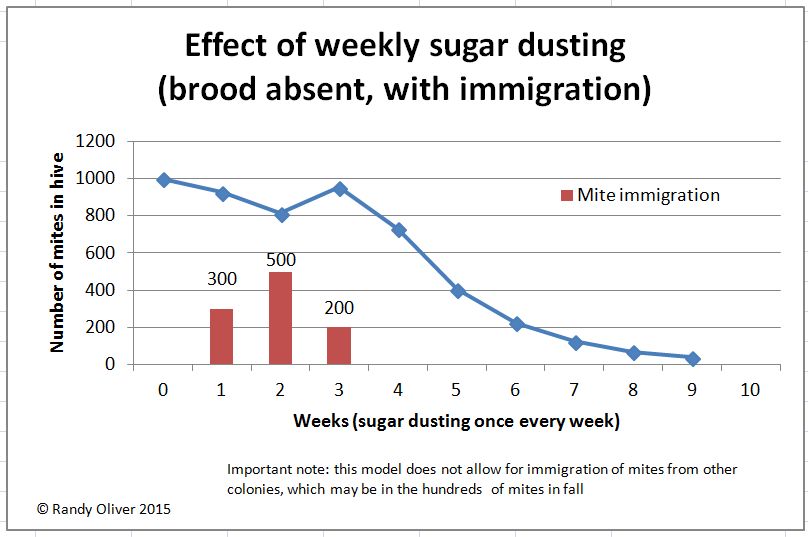
I was recently asked to produce a graph of the expected effect of weekly sugar dusting on the varroa population, so I created an interactive spreadsheet to do so. You can download it here: Dusting simulation spreadsheet.
Here are snips of a couple of simulations:
As you can see, weekly sugar dusting would not be expected to substantially drop the mite population during the broodrearing season–at best it would hold it steady. However, at times when there is no brood present, weekly dusting would be expected to accelerate the decline of the mite population substantially.
More on dust applicators
I originally experimented with top dusting since Dr. Fakhimzadeh found “direct” dusting to be far more efficacious than “aerated” dusting (Fakhimzadeh 2001). However, as easy as it is to top dust, blow dusting may have the advantage of better sugar distribution within the hive. In my previous article I showed a photo of some hand-operated agricultural dusters sold in Europe. Joe Carson of Alaska travels widely in Europe, and is now importing the dusters, to be distributed through Dadant. He reported to me (in part):
The powered sugar sprayers are found all over Europe in the bee supply stores as the preferred method of powered sugar application. Even in the smallest villages in Croatia, I found them, and visited many apiaries whose beekeepers spoke very highly of them.…There are no real scientific tests documented but everyone told me they averaged at least 75 to 80% mite drop when used properly. It takes a time or two to get the hang of it. I have used them for two seasons now. Excellent results with the open or screened bottom board. I slide the non sticky board into the back of the bottom board, lift the lid, prop it open with a stick, and operate the bellows enough pumps to apply powered sugar until it poofs or billows out the top. If the tube with the flared end is inserted too far into the bottom opening, the powdered sugar blows against the back of the hive and is not as effective as when you just barely insert it into the front of the entrance. It takes me about 8 -10 seconds to apply one cup or less of powered sugar per hive. I find that you do not need quite a full cup per colony. I have seen 80 to 85% drop in my tests on my own hives when applied correctly and with a screened bottom board [I asked Joe–he said that he did full colony sacrifice and wash to determine]. I have also used it as a top blower with a screen set on top of the hive. I prefer no insert in the bottom board when applying the powered sugar with the blower from the top. I found I was able to get better coverage with the blower than dumping with a cup and bee brush. It actually blows the powered sugar around the frames and sticks all over the bees and frames. I recommend applying powered sugar with the blowers at least two times and preferably three times in the spring and again in the fall….I do not use any chemicals and my hives winter very well – average 4.5% winter loss in temps as low as -62 F.
More bellows dusters from Europe. The flat ends can be inserted into the hive entrance or under the lid (photos courtesy Joe Carson).
Dusting with half the amount of sugar, and with higher efficacy sounds exciting to me, especially if the higher efficacy translates to more substantial mite control. I plan to try one of Joe’s dusters next season, and perhaps to build a compressor-driven model to rapidly treat large yards by the blow down method described. I’d appreciate hearing reports from anyone else who has tried anything similar.
Update: I was unimpressed with the bellows dusters.
Dusting summary
Please note that the results of these tests are applicable to the top-dusting method described at my website, and may be different for other application methods. Sugar dusting clearly causes a rapid drop of mites in a predictable pattern—most mites fall in the first hour, then the rate tapers off until it returns to baseline in about 24 hours. The method is understandably quicker, and likely more effective, in singles than in doubles.
The acceleration of mite drop is astounding! In the first test, the across the board 10-minute average mite drop (110 mites) was more than double the average 24-hour natural mite fall (48 mites). Whether or not sugar dusting is an effective mite management method, it is clearly a great way to quickly determine the level of mite infestation of a colony. Its great advantage is that it samples virtually all the bees in a colony, as opposed to just a subset.
Sugar dusting as a sampling method is quick and easy, provided that you have screened bottoms or room for an insert. Plus, the mere act of testing removes enough mites to have an effect upon their reproduction. Pettis (2004) and Aliano (2005) found it to be generally safe to the brood, and Dr. Amanda Ellis (pers comm) did not find regular dusting to have any adverse effect upon the colony.
The downsides are that dusting does cause some colony disruption, can’t be done in rain or strong wind, and that at least top-dusting is more effective when applied to singles. In some areas, I’ve heard reports of associated ant problems at the spilled dust.
Sugar dusting utilizes a low-tech physical mode of action against the mite, for which I wouldn’t expect resistance to arise. It is non contaminating and safe to both the operator and the environment. Unfortunately, due to lack of efficacy, I can’t recommend top dusting as a sole means of mite control, but I find it to be a useful tool in my overall mite management.
UPDATE Feb 2009
A new paper on dusting has been published: Amanda M. Ellis, Gerry W. Hayes, and James D. Ellis (2009) The efficacy of dusting honey bee colonies with powdered sugar to reduce varroa mite populations. Journal of Apicultural Research Vol. 48 (1) pp. 72 – 76
“The objective of our study was to determine the efficacy of powdered sugar as a varroa control by comparing mite populations, adult bee populations, and brood area in untreated colonies with those in colonies dusted every two weeks for 11 months with 120 g powdered sugar per application. We found that dusting colonies with powdered sugar did not significantly affect the adult bee population. Within the limits of our study and at the application rates used, we did not find that dusting colonies with powdered sugar afforded significant varroa control.”
I’ve been corresponding with the authors. The key words are “within the limits of our study” and “significant.”
Someone could look at their Figure 4 and come to the conclusion that sugar dusting was very effective, since after a year’s treatment, the dusted colonies had less than a third the mites that they started with, whereas the control colonies returned to the original mite levels. However, the differences were not *statistically* different (although when looking at the standard errors I couldn’t understand why). Unfortunately, the authors declined to share the original data with me to analyze independently.
Note also that this trial was held in Florida, and the mite population dynamics were very different during this trial than those for most temperate areas. Note that the mite populations in both the test and control groups dropped during the summer, which is the opposite of what happens in most temperate areas. This factor alone made any test of efficacy difficult.
This was an excellent and meticulous trial, but it may not really tell us anything new beyond what I have already posted to my website. Top dusting with powdered sugar at the frequency they used (every two weeks), during periods of brood rearing will decrease mite population increase to some extent, but may not drop the level below the initial level (although their Fig 4 shows a substantial initial drop).
Sugar dusting is a useful tool in mite management, and may well see improved application methods that will result in greater efficacy, but it is not a panacea.
Update 10/19/09
Excerpts from an article by Dr Dewey Caron in Beeline
Does this mean PS should not be used in whole colony dustings? In the October 09 in Amer. Bee Jour., Jerry Hayes, one of the authors of the FL study, in his monthly Q&A column answered a letter from Janet Brisson of CA on whether PS works or not as follows:
“Yes research results showed that varroa was not significantly controlled by dusting with
PS. Within the parameters of their research, PS didn’t work great. But that does not mean
that if the research trial were re-structured to treat more often or in a different way that
results would not be different….research shows it doesn’t work long term. But, in real world
situations it does. What to do? I’d treat with PS until research catches up with reality.”
Many beekeepers (including Tom Dowda of FL) dusting whole colonies with PS however believe it is an effective control method. In 9 years of surveying backyard beekeepers in the mid-Atlantic states, I found a growing number of beekeepers using PS for mite control while; over 50% of beekeepers who indicated they were monitoring mite levels prefer to use the PS roll method to determine mite levels vs sticky boards or other methods. In a recent survey of 100 Oregon backyarders (who filled out a survey form during April local association meetings), 65% of the beekeepers who had no over winter losses indicated they used powdered sugar as a control scheme in contrast to 38.5% of beekeeper who did have winter loss (57 of the 100 had a loss; 43 had no loss). Overall colony loss levels were similar for Oregon ( 25.8%) and Mid-Atlantic beekeepers (24 %).
Morris Ostofsky of Eugene OR is one backyard beekeeper who feels PS is effective. He shared with me his success using PS:
“I have successfully used PS to control mites. To give you an example
of the efficacy of the PS treatment here are the results of my
treatment of one of my hives, # 9 – 6, this year. This hive has two
full depth brood boxes.
Date: 8/27 200 mites 24 hr drop using sticky board
8/28 PS dusted
8/31 144 mites on 24 hr on sticky board
9/3 & 9/7 PS dusted
9/10 48 mites on 24 hr sticky board
9/11 & 9/15 PS dusted
9/20 4 mites on 24 hr sticky board
Please note that I would NOT normally treat this often; however,
it was late in the season and I wanted to get the mite count down
before fall. I know this was awfully invasive; however, I felt that
the alternatives were worse. I have used this strategy to reduce
mite loads in other problems hives this year and in previous years.
The fact that I did not lose any hives last winter and had used
powdered sugar to reduce mite counts in fall is an indication that
dusting is effective.
Analysis by Randy
I’m curious about Morris’ results. I’ve done exactly the same thing–dusting at 4 days for two weeks, yet didn’t get anywhere near the results that he got. This was disappointing, since I expected 4-day dustings to drop mite levels substantially.
Since his colony had a high infection, there should have been lots of mites emerging from the brood for at least 12 days (the 9/10 drop would be the last expected large drop, which was fairly large). I’m surprised at how low his 9/20 drop is.
I will try again to see if I can replicate.
————————————————————–
Dear Randy,
I’d like to share my success story with you. I am a hobby beekeeper with 5 hives. I dusted them almost weekly, whenever I open them, all summer. My results matched your observations – it seemed to hold the mites at bay, but there was a spike in the number of mites in August.
Then I said to myself, “the formic acid people only promise a 90% elimination of the mites, you have to leave the pad in place for a certain period of time, and you do it only once it in fall and once in spring. Why not similarly go all-out with the powdered sugar?”
Thus, I chose a week forecasted to have sunny weather the entire week, and powdered my bees once DAILY. I always apply the sugar like you recommend, and I use a dry white board to count the mite fall after an hour, like you. There were over 200 mites each of the first couple days, then the number began to diminish, and by the end of the week when the number of mites was in the teens, I felt I must have reached the 90% treatment level of formic acid, and quit.
My point is this: You have tested everything else. Perhaps short-term DAILY treatments would actually turn out to be a control method.
Thanks!
Jeanne Hansen
Thanks Jeanne,
Daily dusting is actually not necessary, as when a female mite emerges, she spends generally 5-7 days in the phoretic stage before returning to the protection of a brood cell.
So dusting every 3 days should be adequate to knock down most of the mites. Such treatment would need to be continued for about 3 weeks. Morris’s data suggests that this is very effective. If you run such a test, please let me know your results!
Randy
Jeanne replies: Excuse me for arguing, but I thought you said a single dusting only removes 30%-50% of the mites. I don’t understand then, why wait three days to dust again? I’m not talking about the middle of the summer, when new mites are continually emerging with new bees. I’m talking about a fall treatment, similar to a formic acid fall treatment. Let’s dust 5 days in a row and be done with it.
R: Sorry, misunderstood, thinking that you still had brood! Jeanne’s data follows:
OK. Here is my data: The bees were in 3 shallows used for the brood nest. Each box had a nice, large oval pattern of bees on the top bars. I dusted 1/2 C of powdered sugar onto each box = 1 C per deep, and counted the mites after 1 hour.
July 10 – 79 mites
July 20 – 61 mites
July 25 – 30 mites
July 31 – 80 mites
Aug 8 – 74 mites
Aug 24- 125 mites
Aug 30 – 91 mites
Sept 9 – 122 mites
Sept 20 – 252 mites oh, no! what happened?? (rhetorical question) All the above was simply observation to see how many mites the bees had. Then I decided to try and eliminate the mites, as follows, still using 1/2 c sugar for each shallow and waiting an hour to count the mites on the dry white board. [Randy’s note: high mite drops in Sept are normal. Supposing that Jeanne dropped 1/6 of total mites (likely higher since mites move out of the brood at this time), then 252 mites x 6 =about 1500 est. total mites in the colony. For a strong colony in Sept, that is not a particularly worrisome figure at this time of year]
Oct 11 – LOTS of mites, more than 250, so I dusted a second time and after waiting an hour there were EVEN MORE, dusted a third time and ran! The bees were not happy, no count the third time. Temp 54 deg.
Oct 17 – 250 mites. I began leaving the white board in place to see the mite-fall over night. Temp 55
Oct 18 – before dusting there were greater than 250 mites from over night. After dusting, 112 mites only. Temp 58
Oct 19 – before dusting there were mites only under the center of the cluster. After dusting, 56 mites. Temp 60
Oct 20 – before dusting only 100 mites, after dusting, 33. Temp 60 but weather was damp.
Oct 21 – before dusting, 15 mites. I decided not to dust for only 15 mites.
Oct 25 – 27 mites. Temp 70 deg. (Since it was so sunny and warm so late it the year for Wisconsin, I dusted one last time.)
Nov 7 – Since the temperature was a record setting 75 deg, I took the hive totally apart and there was NO brood. I didn’t dust.
Nov 13 – 9 mites Temp 60 deg After a further 24 hours of waiting, only 8 mites more.
Let’s see what my infestation rate was for Oct 25. Eighteen of my shallow frames = 9 of your deeps x 3000 bees/frame =27,000 bees [Randy’s note–about 2000 bees on a deep frame, but close enough]. Let’s say that 27 mites on the white board means 100 mites still on the bees. 100 mites on 27,000 bees = 0.37% To me that means I have controlled the mites with powdered sugar as well as I could have done with formic acid.
The way you and your son can dust 36 colonies in 10 minutes, I should think daily sugar dusting for 5 days would be a do-able control method, even for side liners. After all, they wouldn’t have to stop to count, just dust and go.
Thanks!
Jeanne Hansen
Thank you Jeanne! For all, note that Jeanne’s mite levels built slowly despite roughly weekly dustings during summer. Her idea of cleaning up a broodless colony with daily dustings for a few days sounds great! Plus, theres’ no brood to worry about killing with sugar dust. If any of you try this, please let me know.
References
Aliano, N and MD Ellis (2005) Only large amounts of powdered sugar applied directly to brood cells harms immature honey bees. J Apic Res 44(1): 33-35.
Fakhimzadeh, K (2001) Detection of major mite pests of Apis mellifera and development of non-chemical control of varroasis. Available at http://www.culturaapicola.com.ar/apuntes/sanidad/32_sanidad_varroa_deteccion_control.pdf
Fuchs, S (1985) Untersuchungen zur quantitativen abschätzung des befalls von bienenvölkern mit Varroa jacobsoni Oudemans und zur berteilung des parasiten im bienfolk. Apidologie 16(4):343-368
Pettis, JS, J Kochansky and MF Feldlaufer (2004) Larval Apis mellifera L. (Hymenoptera: Apidae) mortality after topical application of antibiotics and dusts. J Econ Entomol 97(2): 171-176.