The Varroa Problem: Part 17b – The Evolution of Bees, Mites, and DWV
October 28, 2018
Contents
A Primer on the Drivers of Evolution. 2
The Honey bee Populations in the U.S. 4
The Varroa Problem: Part 17b
The Evolution of Bees, Mites, and DWV
First Published in ABJ August 2018
Randy Oliver
ScientificBeekeeping.com
My hope is to clarify exactly how various beekeeping practices affect the evolutionary pressure on the genomes of the honey bee and the varroa/DWV Monster. Armed with such knowledge, every beekeeper can then decide for themselves whether they are contributing to The Solution.
In this age of polarized online discourse, it’s not hard to find those with very strong opinions as to exactly how one should or shouldn’t keep bees, along with some heavy laying on of guilt. The problem is, that often a proponent of a view may have made up their mind without fully understanding the biology involved, but despite that, engage in argument solely to win, as opposed to arguing to learn [[1]]. In this article, I’d like to simply lay out the facts, so at least such arguments can be biologically informed.
I’ve previously explained how we beekeepers are part and party to creating the varroa/DWV “Monster.” We’ve helped it to evolve–by setting up a situation that confers a “fitness” advantage to symbiotic combinations of varroa and DWV that cause the collapse of their host colony late in the season.
The Solution is to strive for two objectives:
- To shift the genomes of our bee populations towards those that exhibit resistance to varroa, and
- To engage in beekeeping practices that eliminate the fitness benefit to the varroa/DWV Monster derived from causing the collapse of their host colony.
Both of the above objectives require a clear understanding of how our actions must be considered in the light of their evolutionary effect upon the bee, the mite, and the virus. So let’s start with…
A Primer on the Drivers of Evolution
First we need to understand the concepts of ecological niche, fitness cost or benefit, and the process of evolutionary genetic adaptation to a niche. Perhaps the easiest way to explain them is to think of the genetic code of a species as being a written business model. Any successful business model represents a way to exploit an available niche in the business environment. Those business models that exhibit the most success (fitness) in that niche succeed; the rest fail.
But as the economic environment tends to change over time, businesses must adapt—that’s evolution. They evolve by tweaking the codes of their business models, and then testing each tweak, by trial and error, in the market (the environment). The best compromises between benefits vs. costs tend to be more successful. We see the resulting evolution of businesses (and species) in retrospect—only the winners survive. And the winners pass the secrets of their success to following generations via their business model (spelled out in the genetic code of a species).
Practical application: evolution is nothing more than the tweaking of a species’ business model over time. This is most clearly exemplified by viruses, which are essentially nothing more than a set of instructions. In this age of molecular biology, evolution is defined as a proportional shift of the genes (alleles) in the overall genetic code of a species’ breeding population [[2]]. So what we need to focus upon are whether our beekeeping practices confer a fitness cost or benefit upon the genes of the breeding populations of the species involved in The Varroa Problem.
There are four species directly involved in The Varroa Problem—the bee, the mite, DWV [[3]], and human beekeepers. In the unmanaged breeding populations of Apis mellifera, natural selection is taking its course—favoring bees that somehow deal with varroa and DWV. What we beekeepers need to focus upon is how our practices either reward or punish virulent varroa/DWV combinations in the artificial niche that we create for the honey bee.
The Niches of the Players
The Niche of the honey bee
The fundamental niche of the honey bee is to exploit the floral resources of any habitat that has adequate nectar- and pollen-producing plants, water, and some sort of nesting cavities. Apis mellifera as a species can adapt to a wide range of biotic (such as plant species and phenology [[4]], competitors, predators, parasites) and abiotic (such as winter temperatures, rainfall or drought) conditions.
Thus, regional populations of A. mellifera–in response to evolutionary selective pressure for overall fitness at exploiting specific environments–resulted in the evolution of the various honey bee subspecies (races) that we know today. Each of those races has adapted to exploit a specific less-than-optimal realized niche (Fig. 1).
Figure 1. The realized niche being exploited by this wild-type colony in Utah is a far cry from optimal for the honey bee. Beekeeper Jerry Shue (who took the photo) and associates are working with their local wild-type population.
Now here’s where it gets interesting: adapting to a more refined realized niche involves tradeoffs—adaptation is always an evolutionary compromise. Those tradeoffs become more apparent during periods of environmental stress (e.g., weather, resources, or parasites). Thus, a generalist bee race may do well in any environment during good times (or if helped by the beekeeper), but a specialized race may outperform the generalist during times of environmental stress.
Practical application: a generic domestic bee stock can perform quite well in good times; a more specialized locally-adapted race may exhibit greater fitness during bad times (like after the honey flow). The question to any breeder is whether to select for colonies that perform best during good times or bad times.
The addition of varroa changed the realized niche of the honey bee—life got much tougher. And when DWV teamed up to collaborate with varroa, life for the bees got tougher yet. Our bees will likely need to make some compromises in order to deal with the mite. Luckily, those compromises do not necessarily involve the bees being more defensive or less productive—but we as an industry need to start putting more selective pressure upon our bees to take over the job of varroa management from us.
Practical application: in the process of breeding generic domesticated bees for traits favored by beekeepers, the tradeoff may be the loss of attributes which confer greater fitness under the unforgiving hand of Nature in the regional niche to which local races are better adapted.
The Niche of The Varroa Mite
In colonies of its native host, Apis cerana, life for varroa is tough. The bees fervently groom the mite from their bodies, only allow it to reproduce in the drone brood, and even then only if it doesn’t harm the pupa. The bee further restricts mite reproduction by only rearing drones from time to time.
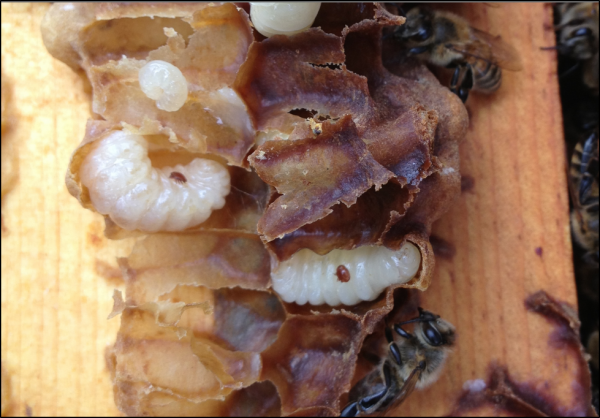
Figure 2. If varroa were able to only exploit the drone brood in Apis mellifera, it wouldn’t be nearly the problem that it is for our bees. The photo above shows mites in drone brood shortly after almond pollination.
Unlike the generalist niche of Apis mellifera, in its original host, Varroa destructor exploits a very narrow niche, each strain of the mite apparently being specifically adapted to reproduce solely upon the drone brood of a regional race of Apis cerana [[5]].
When the Korea strain of varroa adapted to start reproducing in Apis mellifera worker brood, that opened up a whole new niche to exploit—unlimited food, nearly year round, and with very little push back from the bees. The Korea strain of varroa quickly exploited this unoccupied niche, and dispersed throughout the world. It later hooked up with DWV, which allowed it to reproduce [[6]] and disperse even more effectively.
Human perpetuation of non-resistant strains of bees, kept in continually-restocked apiaries, as well as the extra-large broodnests of managed hives, create an even more amendable niche for this strain of varroa. The mite is continuing to adapt to its new host and its symbiotic virus strains [[7]], and has now perfected the technique of killing colonies at just the right time for maximum dispersal of its bloodlines.
The Niche of Deformed Wing Virus
This formerly obscure insect virus rapidly adapted to using a novel vector—varroa—to spread from bee to bee and hive to hive [[8]]. Varroa opened up a whole new niche for the virus, since the mite allowed the virus to bypass the bees’ well-established anti-viral defenses by transmitting the virus directly into the bloodstream of the bee. And it may well be that varroa further helps the virus by digesting the bees’ critical fat bodies (which are involved in numerous aspects of bee physiology and immune response) [[9]].
And then some strains of virus adapted to not only infect individual bees, but to completely overwhelm the colony, causing collapse and the resulting robbing and drift of bees, which allowed for rapid dispersal of those virus strains to colonies throughout the landscape [[10]].
Practical application: we are never going to eliminate varroa or DWV from beekeeping—they are here to stay. If we stay our current course, it’s quite possible that the varroa/DWV Monster will evolve into an even more formidable parasitic combination. On the other hand, we do have the option of adopting management methods that apply selective pressures to tweak the gene pools of the breeding populations of the bees, the mite, and the virus in our favor.
The Breeding Population
A population is made up of members of the same species that interbreed and live in the same area at the same time. The collective set of all the alleles that exist within a population is referred to as its gene pool. Changes in the gene pool pave the way for species adaptation… and, ultimately, evolution. [[11]]
Practical application: if an individual beekeeper wishes to be part of The Solution to The Varroa Problem, they need to keep in mind which breeding population(s) of bees, mites, and viruses their actions affect—or whether they affect them at all.
For example, buying a package of domestic bees and allowing it to die from the varroa/DWV Monster is not going to change the gene pool of the producer’s breeding population in any way—on the contrary—it only increases the proportion of non-resistant bees dispersed that season. And during the collapse of that unfortunate colony, the drift of mites and virulent DWV to other hives only increases the proportion of their genetics in the next generation of your local bee population.
On the other hand, if you’re lucky enough to hive a swarm of gentle bees that turn out to exhibit resistance to varroa, and then produce hundreds of daughters from that queen, you’d have actually helped to shift the genetics of the bee population in a positive way.
Practical application to “treatment-free” beekeepers: simply allowing colonies to die is not part of The Solution—you need to propagate the queen lines that survive.
So let’s take a minute and look at the existing breeding populations of bees in the U.S.
The Honey bee Populations in the U.S.
There’s evidence that there are different regionally adapted “wild-type” breeding populations of the honey bee in the U.S. [[12]], as well as selectively-bred domestic breeding populations of managed bees. Each of these populations is genetically adapted to its specific realized niche—the wild-type bees living without human assistance (or interference); domestic strains adapted to benefit from the large nest cavities and feeding provided by beekeepers. Both of these niches changed with the introduction of varroa as an added biotic factor.
The Wild Type populations: Occupying this “natural” niche (in North America) are established wild-living introduced populations long independent of human management. Prior to varroa–dark, often hot-tempered “wild” honey bees were common in the woods. Some of these populations likely trace back to very early introductions of races of bees from different countries, and although they have likely interbred over the years with other bloodlines, their mitochondrial DNA indicates that they have survived to date without “rescue” by humans [[13]]. These wild populations, due to their diversity and apparent survivorship despite varroa, may offer valuable genetic combinations for improving our managed stocks. This is also true for the more recent invasion by the hybridized Africanized bee.
| Here we get into semantics— a feral animal is one which has escaped captivity or domestication and gone on to survive in the wild without assistance. A feral population is descended from an inbred domesticated stock. This would be the case for colonies founded by escaped swarms from commercial non-resistant bloodlines. These days, most of those feral colonies perish within a year or two from the varroa/DWV Monster.
On the other hand, the early introductions of honey bees to North America likely carried genomes closer to “wild type,” and readily adapted to ecological habitats similar to those of their natal countries. The fact that mitochondrial bloodlines of races of bees not propagated by commercial breeders continue to exist throughout the U.S. indicates that at least some of our free-living honey bee populations can be considered as wild type rather than feral escapees.
|
Some of these wild-type populations are isolated from managed colonies, and may be of low density (as in the Arnot Forest)—which would not reward varroa/DWV symbionts that cause their host colony to collapse (especially if such collapse occurs during the freezing winter when no dispersal can take place).
Of interest is that these wild-type populations of honey bees appear to be able to maintain some degree of genetic integrity despite the presence of managed bees in the landscape [[14]]. However, such wild-type populations at the interface with managed apiaries may face competition from those large-cavity colonies, and need to deal with parasite drift, robbing pressure, and the infusion of genetics through interbreeding with the managed bees.
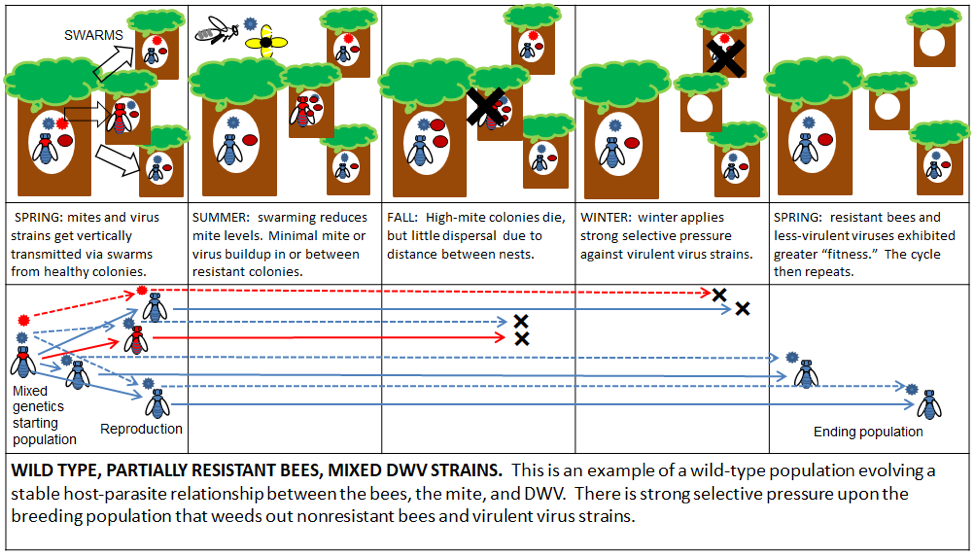
I’ve made up some diagrams to illustrate the effects of various management practices upon bee and DWV evolution. I’ll first offer an idealized version of the natural selective pressure on a wild type population living in a sparsely-populated (by bees) forest (Figs. 3 & 4):
Figure 3. The legend for the diagrams.
Figure 4. The result of natural selective forces upon the evolution of the genomes of the bee and DWV in a wild-type population. Simple interpretation for all diagrams: a left-to-right shift to blue is good, a shift to red is bad [[15]].
Practical application: these naturalized populations of bees don’t need no stinkin’ humans, and are likely better off without us. They would be great candidates for Darwinian beekeeping. However, I for one, enjoy working my own domesticated bee stock without protective gear or the “angry” and exhausting buzz of a cloud of bees around my head. There’s nothing wrong with keeping gentle bees—how many people try to make a living by milking wild goats?
The Domesticated Livestock population: populations of bees in which the genetics are controlled by the queen breeder/producers, who typically select for gentleness and productivity. The niche to which these bees are adapted is very different than that of the wild type bee—Dr. Tom Seeley listed 20 major differences in his article Darwinian Beekeeping [[16]]. The most important differences are:
- The expanded nest size (bigger than the typical bee-occupied tree cavity), which allows for much larger broodnests and colony strengths, especially if the colonies are managed to prevent swarming. The big broodnests create the perfect feeding ground for varroa, allowing the mite to develop extremely large populations in the hives before they collapse.
- Supplemental feeding of colonies, which allows for maintaining apiaries at beyond the carrying capacity of the landscape, resulting in periodic nutritional stress (forcing longer-range foraging), and greater robbing pressure (which favors mite/virus dispersal).
- The high density of colonies—often dozens to hundreds (or thousands) in close proximity. This allows for vastly more efficient colony-to-colony parasite transmission (dispersal), prior to, and during collapse.
- The restocking of parasite-killed hives with fresh colonies, thus removing any fitness disadvantage to parasites from causing the death of their host colony.
- The use of miticides and antibiotics to control parasites, which removes the evolutionary pressure for the bee population to develop natural resistance.
I’m not saying anything critical of the above—I’m a commercial beekeeper myself. As with other forms of livestock husbandry, our current food-production system rewards large-scale livestock operations, and there are business opportunities (niches) for beekeepers who can provide migratory pollination services, produce honey at large-scale, and provide bees and queens for sale to other beekeepers. Professional beekeeping has evolved in order to take advantage of those business opportunities.
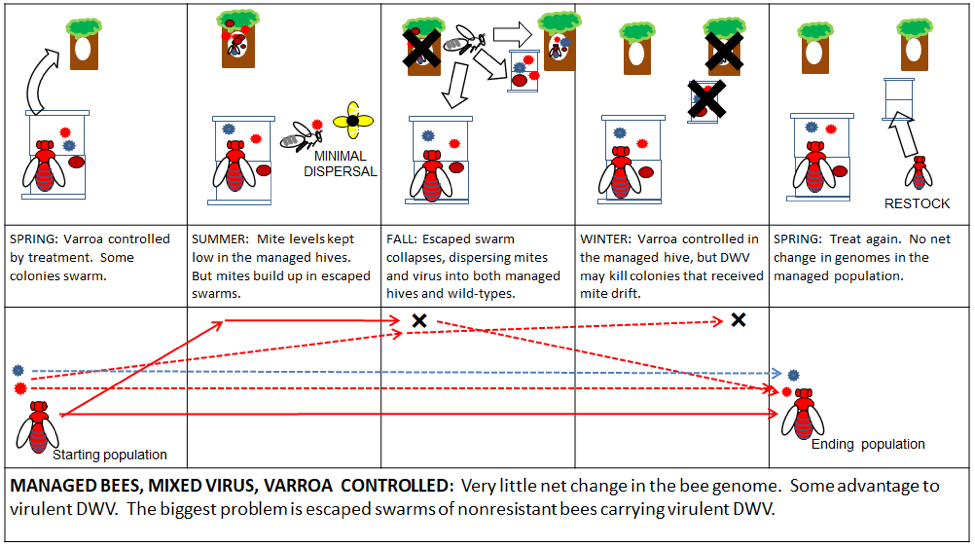
There is little financial incentive for professional beekeepers to change the above practices, since they work (Fig. 5). Accordingly, the queen producers will continue to breed bees specifically adapted to this niche—it is my hope that commercial genetics will soon include traits for varroa resistance. There is nothing “wrong” with commercial bee stock, any more than there is anything “wrong” with domesticated cattle, chicken, or dogs. These animals are bred for a purpose, and the queen breeders do an excellent job at supplying our industry with bees adapted to the livestock niche.
Figure 5. In a well-managed operation of domestic bees, there is little net effect—good or bad—upon bee or virus evolution. However, this scenario is completely dependent upon the availability and application of effective miticides. Compare the above to what happens if those treatments are not effectively applied (Fig. 6).
A key practical note: based upon the data of Mangus and Szalanski [[17]], it appears that these domesticated stocks of bees do not necessarily survive very well in the wild. There is absolutely no reason to expect domesticated bees to perform well in a niche to which they are not well adapted. And currently, most domesticated stocks have not been selected for resistance to the either varroa or DWV.
To effect evolutionary change in any breeding population’s gene pool, there must be a fitness advantage (or cost) conferred by certain alleles in both reproduction and dispersal. This applies to the bee, the mite, and the virus. So let’s look into those two drivers of evolution:
Reproduction and Dispersal
Shifting the gene pool of the honey bee towards mite resistance will require the disproportionate reproduction of resistant queens. Unfortunately, there are currently not enough queens from proven mite-resistant stock available for purchase. One problem is that we don’t have an impartial testing organization to compare purported resistant stocks in various areas
Practical need: our industry could use an independent All America Selection organization to test purported mite-resistant queen stocks in each region of the country. We could copy the established models for testing plant cultivars [[18]]. We’re unlikely to see any serious change in bee genetics until proven mite-resistant stock becomes more readily available to commercial beekeepers.
Commercial queen producers sell over a million queens each season. They are going to produce non-Darwinian bees. But there’s no reason that they couldn’t be producing gentle, productive, mite-resistant stock.
Practical application: be realistic. If you’re in an area with a preponderance of commercial beekeepers, as a small-scale beekeeper, you’re not going to shift the genetics of the breeding population. On the other hand, if there are no large-scale beekeepers around, and you have cooperative recreational beekeepers willing to work with you, by all means try to propagate and disperse mite-resistant bees.
The above is tough if other beekeepers continue to import domestic stock from elsewhere each season—that genetic infusion will make it difficult for any regional stock to develop. My hat is off to those beekeepers worldwide who are working cooperatively to propagate varroa-resistant regional bee stocks. Otherwise, your efforts will likely be futile.
Bottom line: If you want to change the genetics of a bee breeding population, you need to focus upon increasing the reproduction and dispersal of mite-resistant queen lines. If you’re not reproducing a lot of queens yourself [[19]], then get some resistant queens from (or to) a producer who will. And only when those better queens get dispersed throughout your region’s breeding population can mite resistance become a reality.
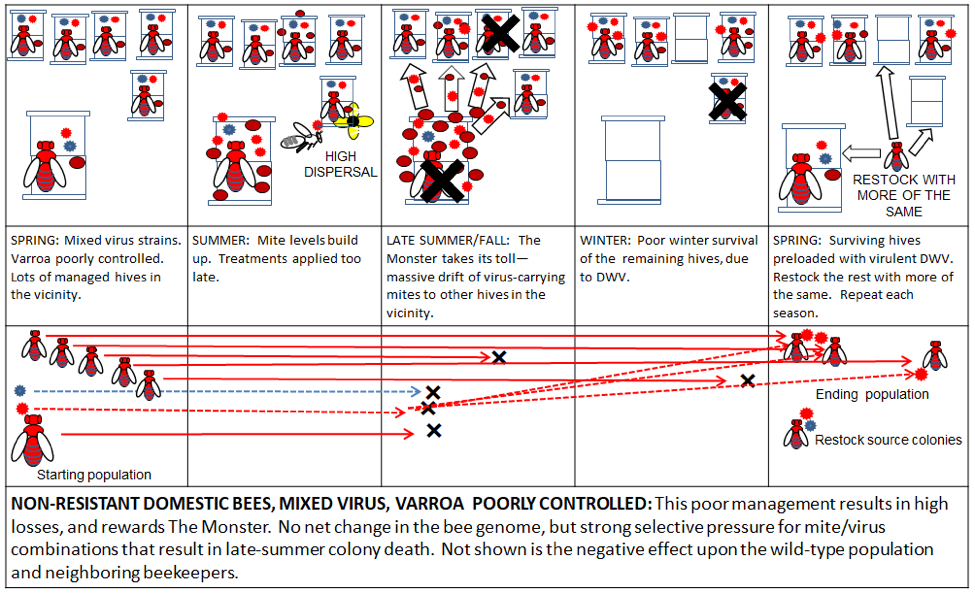
Dispersal of mites and DWV is another matter, and is largely a direct result of our beekeeping practices. Large apiaries, poor mite management, and the replacement of collapsed colonies with more non-resistant bees favor the most virulent symbiotic combinations of varroa and DWV (Fig. 6).
Figure 6. Poor varroa control in a managed apiary using non-resistant domestic stock rewards The Monster, since it confers a fitness advantage to mite/virus combinations that kill their colony in late summer. This not only perpetuates The Varroa Problem, but may even make it worse over time.
Similar to malaria, DWV is generally not much of a problem without an effective vector to disperse it. That effective vector is a high infestation of varroa. And if the virus manages to kill its host colony during robbing season, it then very effectively uses the mite to disperse it to new host hives. Both the virus and the mite evolutionarily benefit from such dispersal.
Practical application: dispersal, dispersal, dispersal—I said it three times. All beekeepers should work to minimize the dispersal of varroa and virulent DWV from their hives to others in the vicinity, as this only rewards The Monster [[20]].
I’ll continue next month with illustrations of the genetic consequences of other forms of apiary management, including “treatment free” beekeeping.
Acknowledgements
Thanks as always to Pete Borst for research assistance, and to all the dedicated and hard-working bee researchers from whose publications I draw useful information.
Notes and Citations
[1] A recent article in Scientific American addresses the difference: The Tribalism of Truth. Available for free download.
[2] An allele is an alternate form of a gene; in the breeding population of any species, there may be several common alleles for any gene (e.g., for black, brown, or white coloration). There are also other heritable characteristics besides genes, such as the epigenetic adjustments to the environmental factors.
[3] Varroa also transmits other viruses, but they don’t appear to have evolved to take advantage of varroa as has DWV—see:
Nazzi, F, et al. (2012) Synergistic parasite-pathogen interactions mediated by host immunity can drive the collapse of honeybee colonies. PLoS Pathog 8(6): e1002735.
Some (such as the paralytic viruses) tend to be self limiting, due to their high virulence in individual bees. But it is DWV alone that has intimately coupled with varroa across the world. This is not to say that other viruses may not do so.
[4] The seasonal timing of bloom of the local plant community.
[5] Dr. Denis Anderson brought this to our attention in 1998, eventually leading to his identifying and renaming Varroa destructor. The following two papers track the process of discovery.
Denis L Anderson & Stefan Fuchs (1998) Two genetically distinct populations of Varroa jacobsoni with contrasting reproductive abilities on Apis mellifera, Journal of Apicultural Research, 37(2): 69-78.
Anderson, D (2006) Clarification of aspects of Varroa reproduction—first stage of a possible new control method. RIRDC Publication No. W06/007. Open access, but unfortunately was not funded.
[6] Di Prisco, G, et al (2016) A mutualistic symbiosis between a parasitic mite and a pathogenic virus undermines honey bee immunity and health. Proceedings of the National Academy of Sciences 113(12): 3203-3208.
[7] DWV is not new to the honey bee, but existed as an opportunistic “cloud” of variants, very rarely causing noticeable problems. But once varroa entered the picture, two variants (DWV A and B) quickly outcompeted the numerous other strains of the virus (see the following citation). So when I refer to the varroa/DWV “Monster,” I’m talking about these varroa-adapted DWV strains.
[8] Martin, SJ, et al (2012) Global honey bee viral landscape altered by a parasitic mite. Science 336: 1304-1306.
[9] Despite varroa and DWV having been a major problem for decades, we are still on the learning curve as to many details of the biology of this complex host-parasite relationship.
[10] Nazzi (2012) op cit.
[11] www.nature.com/scitable/ebooks/essentials-of-genetics-8/contents
[12] https://scientificbeekeeping.com/whats-happening-to-the-bees-part-5-is-there-a-difference-between-domesticated-and-feral-bees/
[13] Mitochondrial DNA can be used to track unbroken maternal bloodlines. Since the maternal bloodlines of some wild-type bees do not exist in the commercially-managed bee populations, then they could not be recently-escaped ferals, and were apparently not extirpated by the invasion of varroa.
[14] Daly, HV, et al (1991) Clinal geographic variation in feral honey bees in California, USA. Apidologie 22: 591-609. Open access. See Figure 3 at my What’s Happening Part 5 link above.
[15] I toyed with a number of ways to illustrate the effects of management practices upon bee and DWV evolution. I’ll post these illustrations to to my website for better viewing.
[16] Seeley, TB (2017) Darwinian Beekeeping: An Evolutionary Approach to Apiculture. ABJ 157(3): 277-282. Open access at https://www.naturalbeekeepingtrust.org/darwinian-beekeeping
[17] Magnus, R and AL Szalanski (2008) Genetic variation in honey bees from south central United States. Poster at 2008 ESA Open access. Compare the pie charts (Figure 4) that I created from their data at my What’s Happening Part 5 link above
[18] https://all-americaselections.org/about/
Another existing program is the Smart Bees project in Europe.
[19] https://scientificbeekeeping.com/queens-for-pennies/ and https://scientificbeekeeping.com/small-scale-queenrearing/
[20] There is the special case with the Bond Method, in which such dispersal puts extremely strong selective pressure upon the bees; but the Bond Method requires that one only restock colonies with survivors. I’ll illustrate this in the next article.